CHEM 2323 Study Guide - Substituent, Alkane, Alkyne
Document Summary
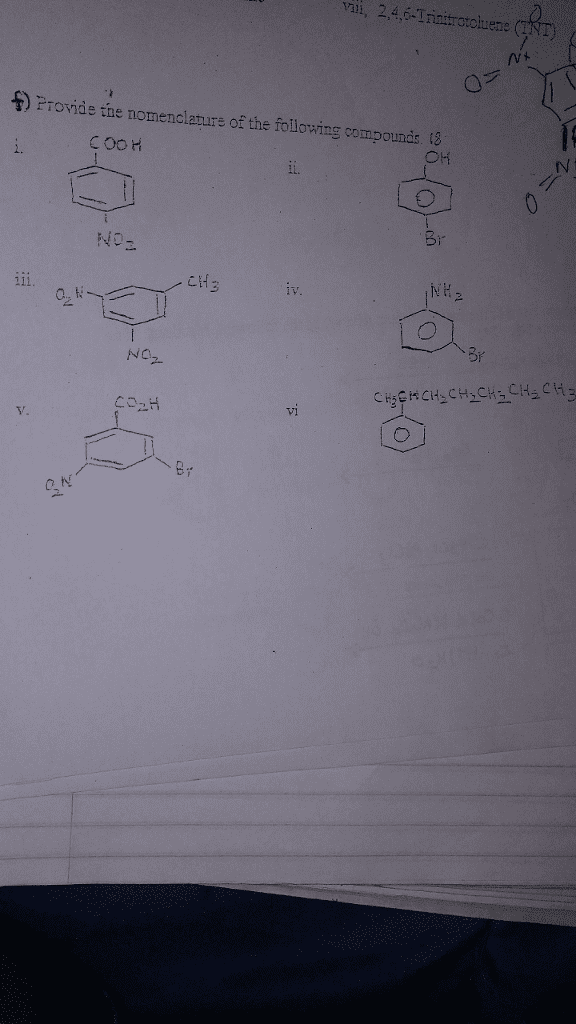
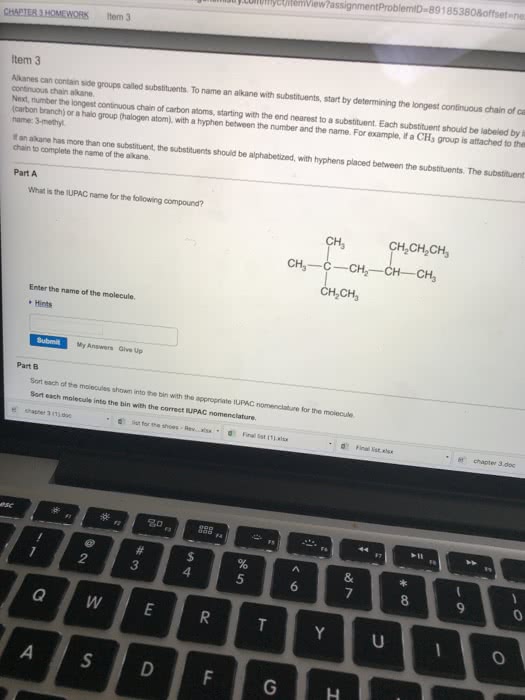
Nomenclature is providing names for any organic chemistry product. When coming across a nomenclature problem (when you are given the compound and have to name it), follow the basic nomenclature rules listed below: find the longest continuous chain of carbons in the compound. This number will be the base name of the compound. If a compound only has 1 substituent, place that at the lowest number possible in the chain. For example if given a central carbon chain of 5, with a substituent placed 1 carbon away from the center one, you would name that substituent in the 2nd carbon, not the 4th carbon: ex: , not: note: name any substituents similarly when naming the central chain. Simple alkane substituents are given yl after the root (if a substituent has 1 carbon attached, the substituent is methyl, 2 carbons = ethyl)