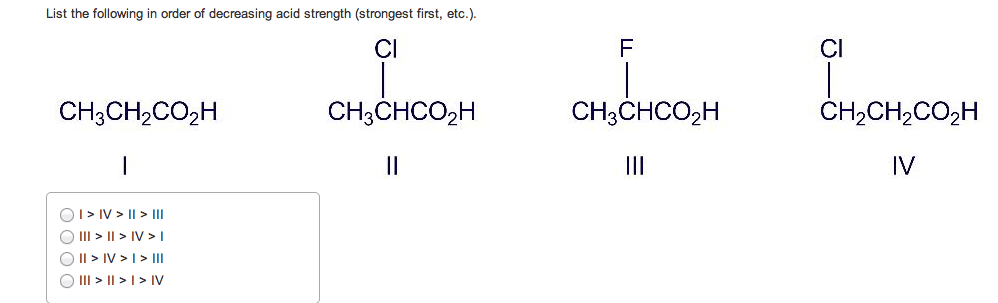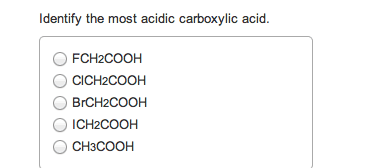CHEM 2323 Study Guide - Conjugate Acid, Organic Chemistry, Elementary Charge
11 views4 pages
Document Summary
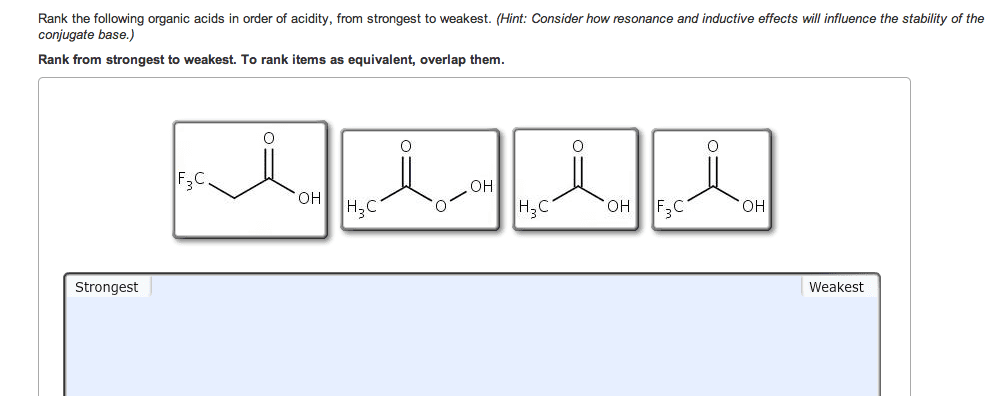
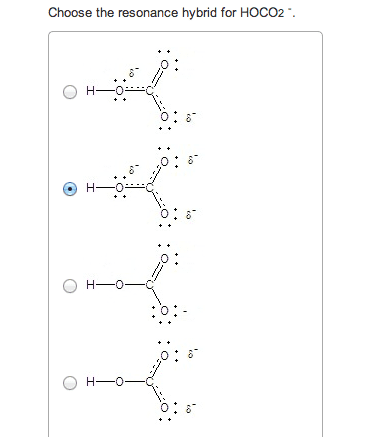
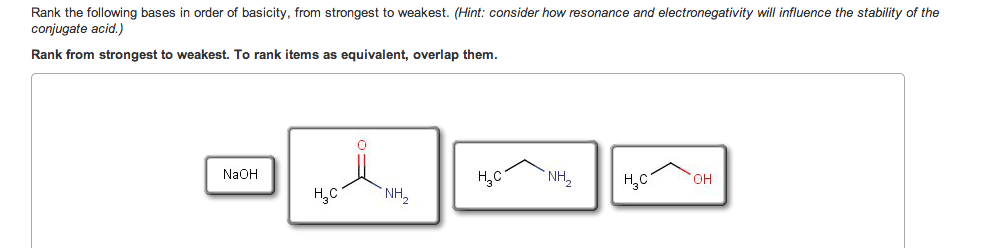
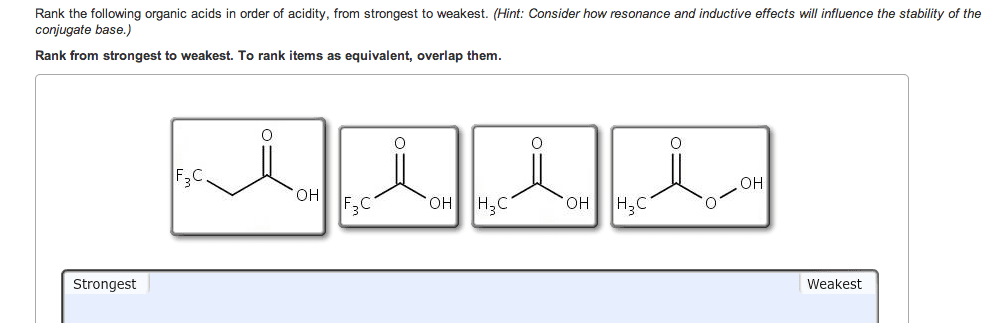
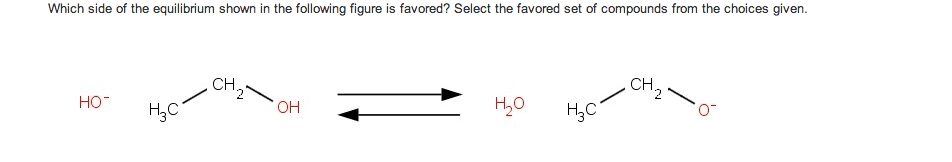
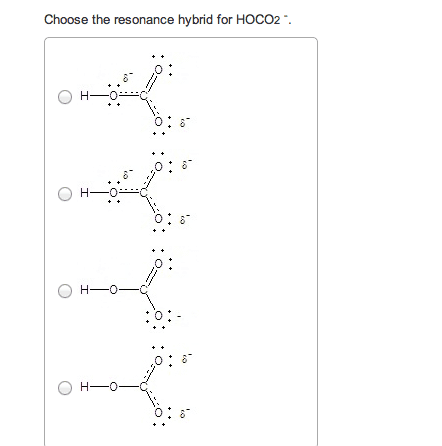
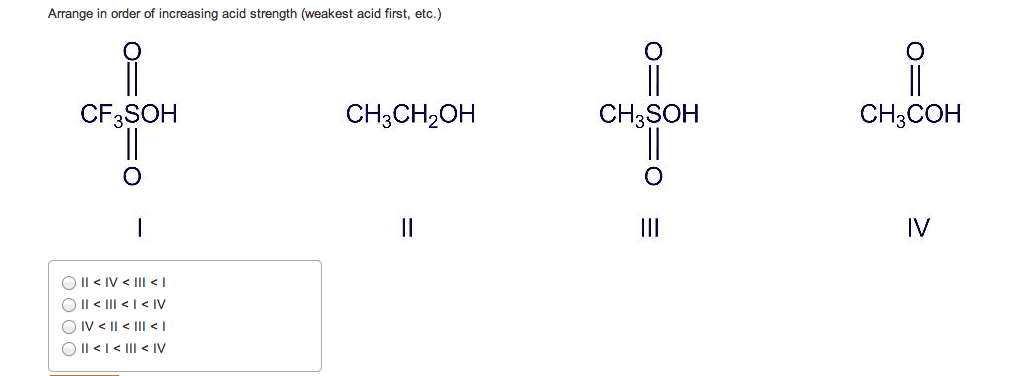
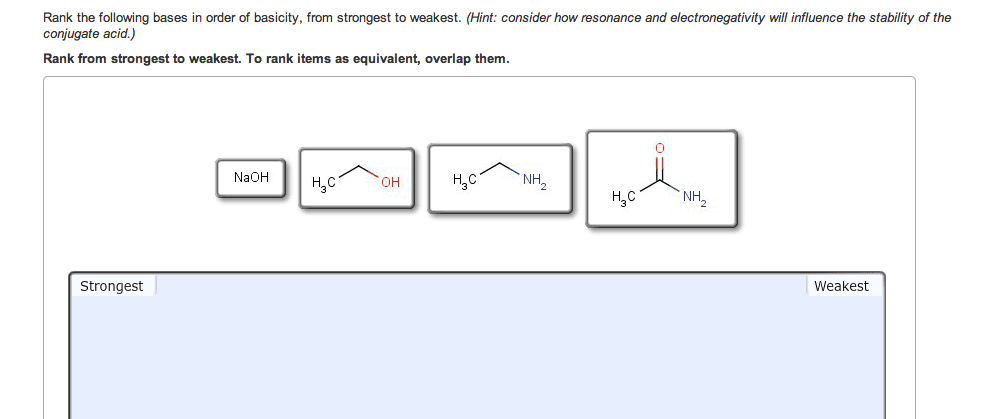
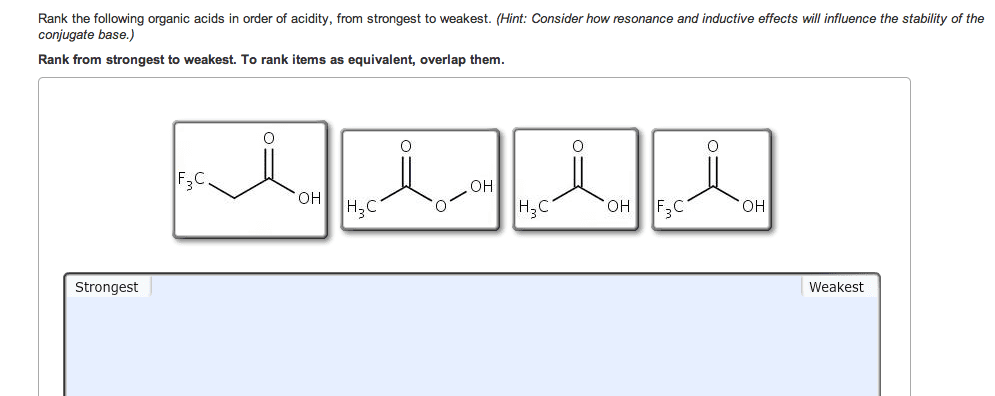
If you remember from general chemistry, there are 3 types of acids: arrhenius, bronsted, and lewis. Also, there are 6 strong acids: hcl, hbr, hi, hno3, h2so4, and hclo4. However, in organic chemistry, we are concerned with bronsted definition: the ability of an acid to donate a proton. What defines how acidic something is in organic chemistry is the stability of the conjugate base formed (which is a carboanion). The more stable the conjugate base is, the more likely that the acid will be willing to donate a proton. There are some general criteria to be considered when determining which substances are more acidic when compared to others: resonance: for organic chemistry, resonance is the biggest contributor to stability of the conjugate base. The more resonance structures that a compound has, the greater the ability that the compound can delocalize the negative charge across the entire compound.
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232