CHEM 2323 Study Guide - Midterm Guide: Pi Bond, Polar Effect, Substituent
Document Summary
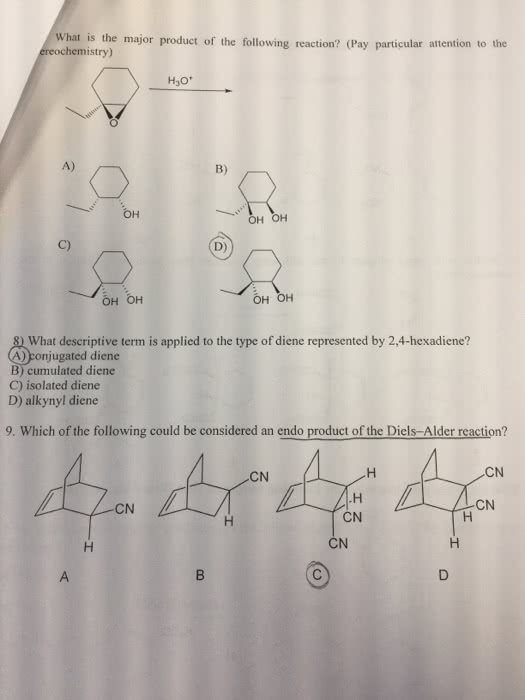
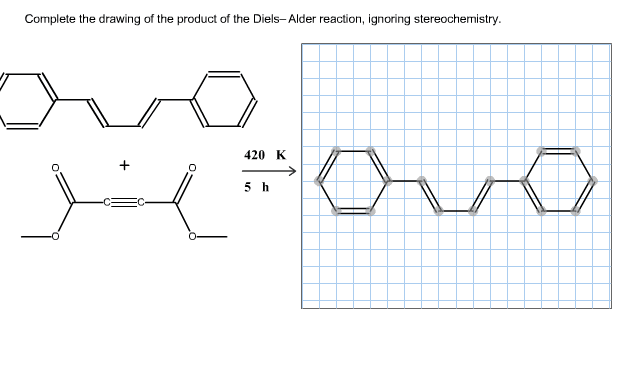
The good thing about ch 15 is that there are very few new reactions: you really only need to know the 1,2/1,4 addition, and the diels alder reaction. However, you must know these reactions in detail! It is important to know everything about diels alder reaction: reaction, mechanism, stereochemistry, regiochemistry, etc. Diels alder reaction is the meat of this chapter, so know it very well! A conjugated diene can extract a hydrogen (from hbr, h+ and h2o, etc ), forming a carbocation intermediate. Notice that the carbocation forms adjacent to a double bond because of resonance, stabilizing the carbocation. If the reaction takes place in cold conditions, or the absence of heat, then the bromine will directly attack the initial carbocation formed, forming the 1,2 product, also known as the kinetic product. If this reaction takes place under heated conditions, then this allows the carbocation to switch places and rearrange onto the first carbon.