CHEM 1212K Lecture Notes - Lecture 11: Sodium Acetate, Conjugate Acid
Document Summary
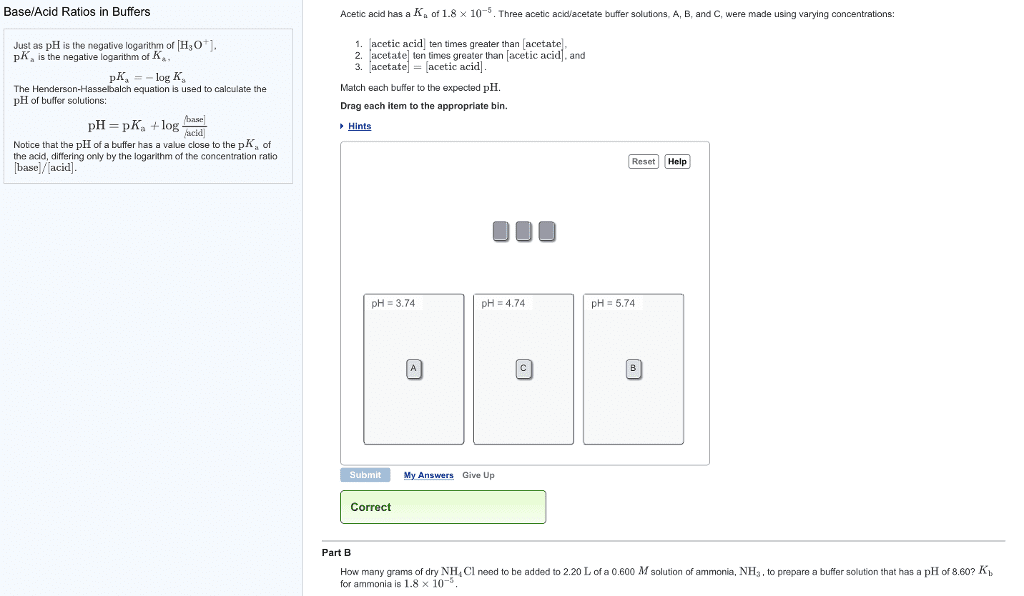
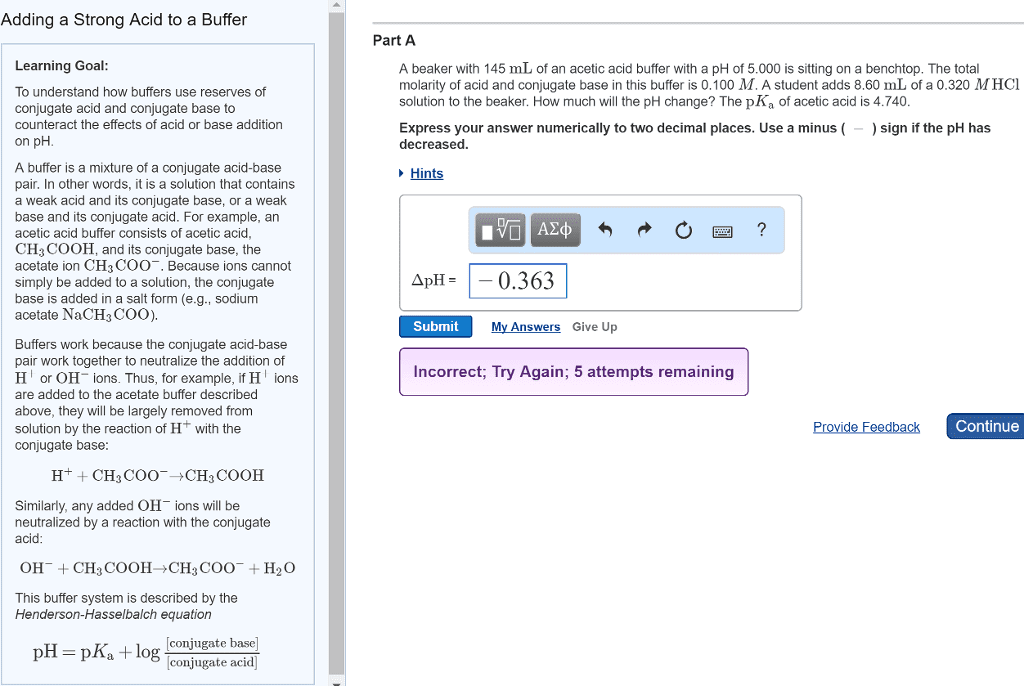
Problems using the henderson-hasselbalch equation: an acid and its salt. A buffer is prepared containing 1. 00 molar acetic acid and 1. 00 molar sodium acetate. We must know the pka of acetic acid. Most times, the problem will provide the pka. If the problem provides the ka, you must convert it to the pka. Be aware, your teacher may force a situation where you must look up the ka. Even in this era of fairly easy internet access, try one of the appendices of your textbook (if you have one). Tables of ka values are a standard feature of most textbooks. The ka of acetic acid is 1. 77 x 10 5 pka = - log ka = - log 1. 77 x 10 5 = 4. 752. Next, we simply insert the appropriate values into the hh equation: x = 4. 752 + log (1. 00 / 1. 00) Since the log of 1 is zero, we have ph = 4. 752.