1
answer
0
watching
935
views
28 Sep 2019
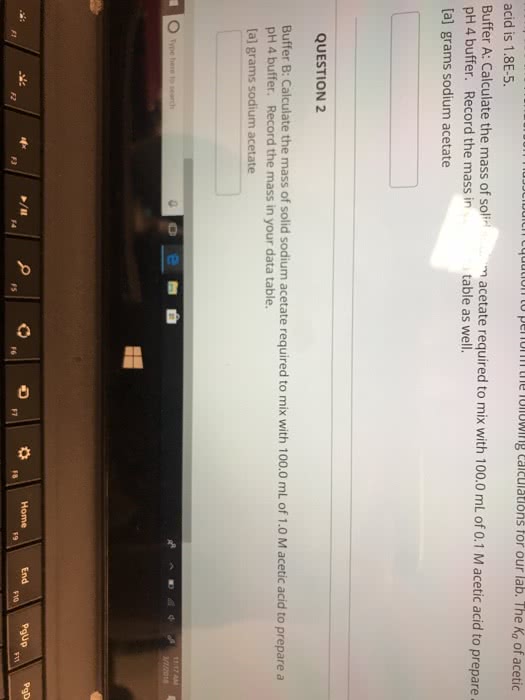
Using the Henderson-Hasselbalch equation, determine the amount of acetic acid (volume in mL, starting with a 1.0 M stock solution) and sodium acetate trihydrate (mass in grams) that must be mixed together to make 100 mL of a 50 mM acetate buffer with pH=5.20
Calculate how much 1.00 M (HCl) and sodium acetate trihydrate must be used to make the buffer described in the above problem if you had access to only solid sodium acetate trihydrate and HCl, not acetic acid.
Using the Henderson-Hasselbalch equation, determine the amount of acetic acid (volume in mL, starting with a 1.0 M stock solution) and sodium acetate trihydrate (mass in grams) that must be mixed together to make 100 mL of a 50 mM acetate buffer with pH=5.20
Calculate how much 1.00 M (HCl) and sodium acetate trihydrate must be used to make the buffer described in the above problem if you had access to only solid sodium acetate trihydrate and HCl, not acetic acid.
Beverley SmithLv2
28 Sep 2019