CHEM 123 Lecture Notes - Lecture 3: Path Dependence, Lead Zirconate Titanate, Heat Capacity
CHEM 123 verified notes
3/4View all
Document Summary
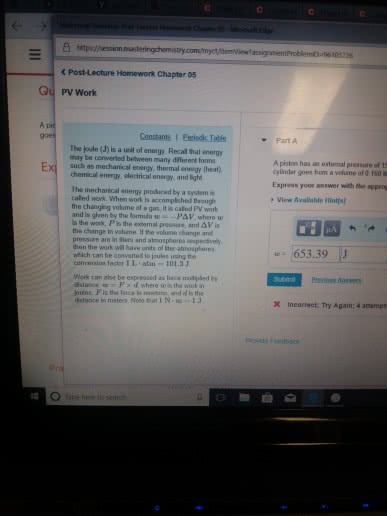
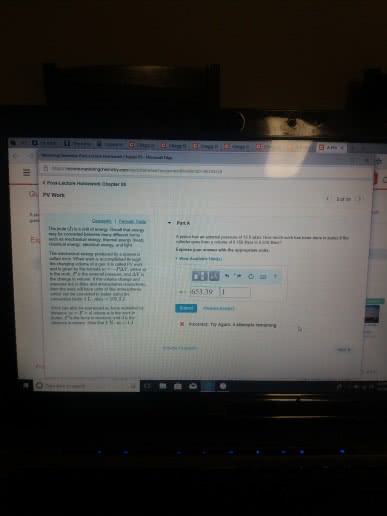
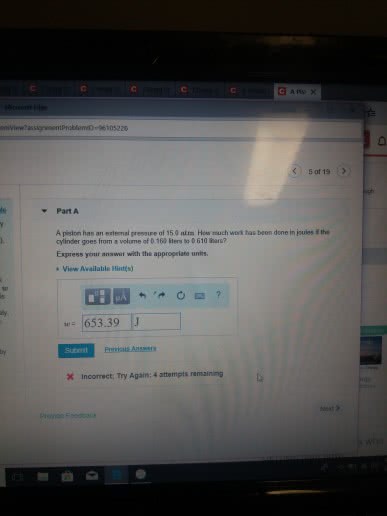
From physics w: d opposing pressure . w. A state function describes the state of a system . It does not depend on how the system came to be in that state. Path variables ( i . e : process variables. The value of path variables depends on how a process is performed , not just its starting and ending. Slow expansion isothermal of an ideal gas ( t , = tz ) expansion of an ideal gas. Ti graph starts once single grain of sand is removed. If q and w are process variables how can. In a jet , why must air from outside at g km altitude be cooled to get a comfortable cabin temperature o. The air must be compressed to get an appropriate pressure. O because fast compression f- w > o because compression. Ov it moves up , pushes piston complicated monitor to heat measure only i work rather than heat eliminate w.