1- The molar heat capacity of silver is 25.35 J/molâ âC. How much energy would it take to raise the temperature of 8.20 g of silver by 14.5 âC?
Express your answer with the appropriate units.
_____________
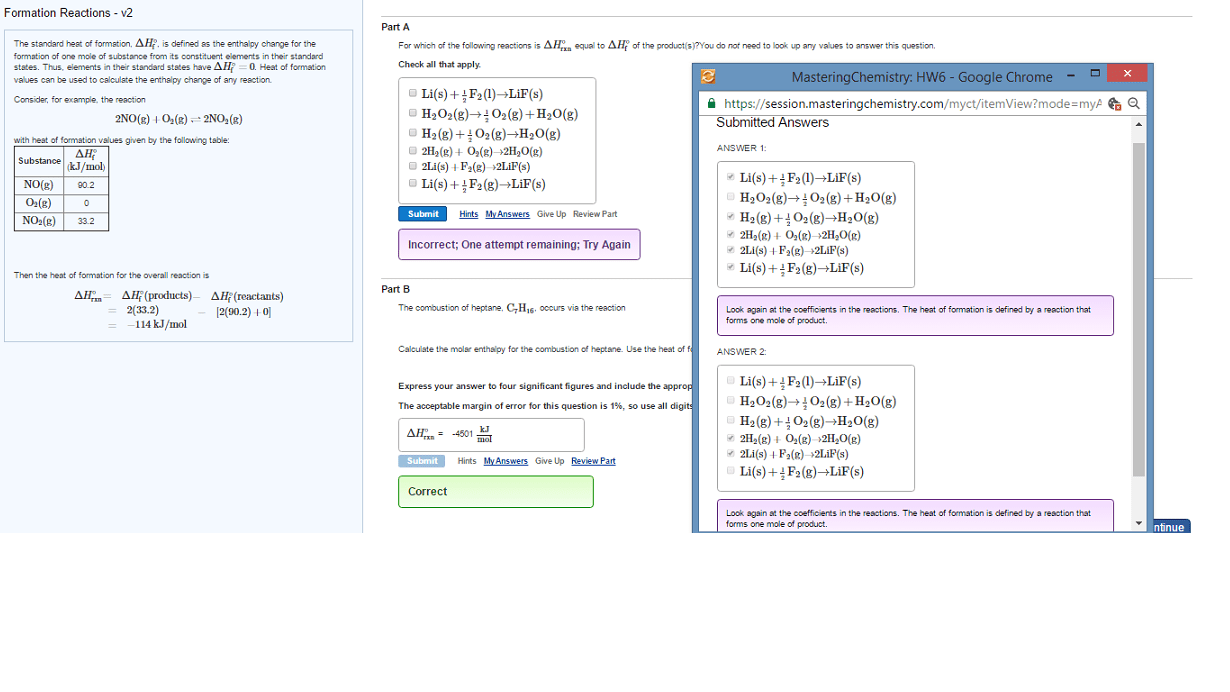
Part A
Calculate the enthalpy of the reaction
2NO(g)+O2(g)â2NO2(g)
given the following reactions and enthalpies of formation:
12N2(g)+O2(g)âNO2(g), ÎHâA=33.2 kJ
12N2(g)+12O2(g)âNO(g), ÎHâB=90.2 kJ
Express your answer with the appropriate units.
2- A total of 2.00 mol of a compound is allowed to react with water in a foam coffee cup and the reaction produces 196 g of solution. The reaction caused the temperature of the solution to rise from 21.00 to 24.70 âC. What is the enthalpy of this reaction? Assume that no heat is lost to the surroundings or to the coffee cup itself and that the specific heat of the solution is the same as that of pure water.
Enter your answer in kilojoules per mole of compound to three significant figures.
_________
3- A volume of 110. mL of H2O is initially at room temperature (22.00 âC). A chilled steel rod at 2.00 âCis placed in the water. If the final temperature of the system is 21.00 âC , what is the mass of the steel bar?
Use the following values:
specific heat of water = 4.18 J/(gâ âC)
specific heat of steel = 0.452 J/(gâ âC)
Express your answer to three significant figures and include the appropriate units.
Part B
The specific heat of water is 4.18 J/(gâ âC). Calculate the molar heat capacity of water.
Express your answer to three significant figures and include the appropriate units.
______-
4- What is the enthalpy for the following reaction?
overall: N2O4â2NO2
Express your answer numerically in kilojoules per mole.
____________
Part B
Calculate the enthalpy of the reaction
4B(s)+3O2(g)â2B2O3(s)
given the following pertinent information:
B2O3(s)+3H2O(g)â3O2(g)+B2H6(g), ÎHâA=+2035 kJ
2B(s)+3H2(g)âB2H6(g), ÎHâB=+36 kJ
H2(g)+12O2(g)âH2O(l), ÎHâC=â285 kJ
H2O(l)âH2O(g), ÎHâD=+44 kJ
Express your answer with the appropriate units.
_____________
5- What is the balanced chemical equation for the reaction used to calculate ÎHâf of MgCO3(s)?
If fractional coefficients are required, enter them as a fraction (i.e. 1/3). Indicate the physical states using the abbreviation (s), (l), or (g) for solid, liquid, or gas, respectively without indicating allotropes. Use (aq) for aqueous solution.
Express your answer as a chemical equation.
__________
6- Part A
Determine the enthalpy for this reaction:
Ca(OH)2(s)+CO2(g)âCaCO3(s)+H2O(l)
Express your answer in kilojoules per mole to one decimal place.
_______
Part B
Consider the reaction
Ca(OH)2(s)âCaO(s)+H2O(l)
with enthalpy of reaction
ÎHrxnâ=65.2kJ/mol
What is the enthalpy of formation of CaO(s)?
Express your answer in kilojoules per mole to one decimal place.
________
7- Part A
Write a balanced chemical equation depicting the formation of one mole of H2O2(g) from its elements in their standard states.
Express your answer as a chemical equation. Identify all of the phases in your answer.
_____
Part B
For H2O2(g), find the value of ÎHâf. (Use Appendix C)
Express your answer using five significant figures.
_____
PART C: Write a balanced chemical equation depicting the formation of one mole of CaCO3(s) from its elements in their standard states.
Express your answer as a chemical equation. Identify all of the phases in your answer.
_____
Part D
For CaCO3(s), find the value of ÎHâf. (Use Appendix C)
Express your answer using five significant figures.
_____
Part E
Write a balanced chemical equation depicting the formation of one mole of POCl3(l) from its elements in their standard states.
Express your answer as a chemical equation. Identify all of the phases in your answer.
____
Part F
For POCl3(l), find the value of ÎHâf. (Use Appendix C)
Express your answer using four significant figures.
_____
Part G
Write a balanced chemical equation depicting the formation of one mole of C2H5OH(l) from its elements in their standard states.
Express your answer as a chemical equation. Identify all of the phases in your answer.
_____
Part H
For C2H5OH(l), find the value of ÎHâf. (Use Appendix C)
Express your answer using four significant figures.