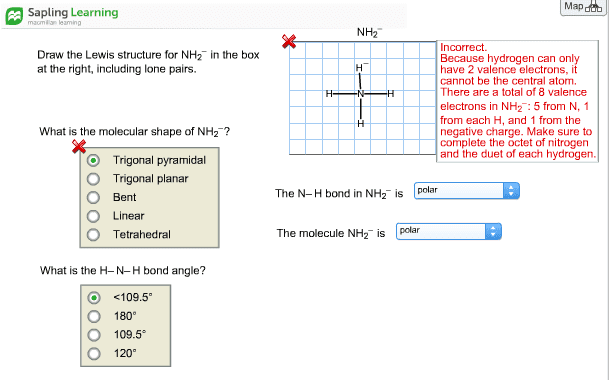
CHEM 101 Chapter Notes - Chapter 8: Trigonal Planar Molecular Geometry, London Dispersion Force, Valence Electron
Document Summary
Charge clouds - electrons in bonds and in lone pairs; repel each other and stay as far away as possible. A single electron, a lone pair of electrons, a single bond, a double bond, and a triple bond each represent one charge cloud. 1) write electron dot structure for molecule and count electron charge clouds surrounding atom of interest. 2) predict geometric arrangement of charge clouds. 3 charge clouds, trigonal planar = 120. 4 charge clouds, trigonal pyramidal = 107. Covalent bond results when two atoms approach each other closely enough so that a singly occupied valence orbital in one atom spatially overlaps a singly occupied valence orbital on the other. Sigma bonds - head on orbital overlap. 2 charge clouds - sp; 3 charge clouds - sp2; 4 charge clouds - sp3. Bond dipole - has a positive and a negative; arrow with a cross at one end ( neg -|--> pos)