CHEM 103 Chapter Notes - Chapter 3: Reagent, Stoichiometry, Minuscule 33
Document Summary
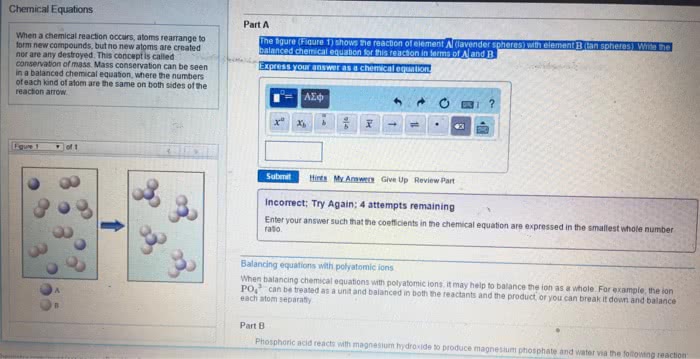
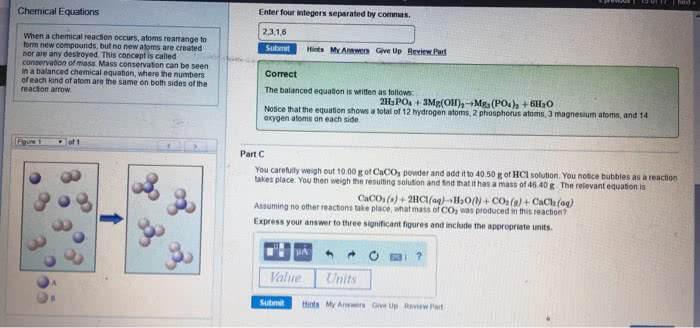
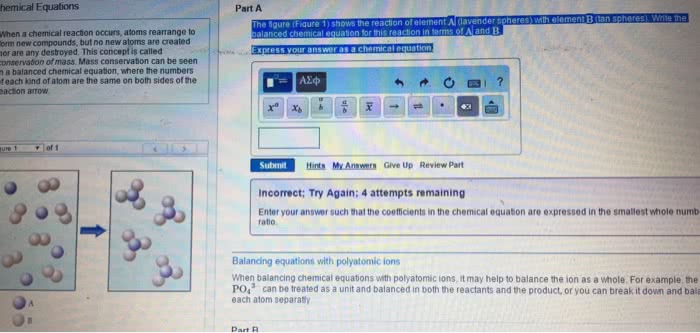
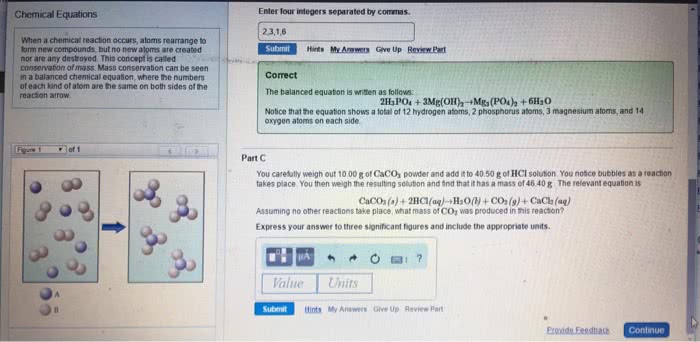
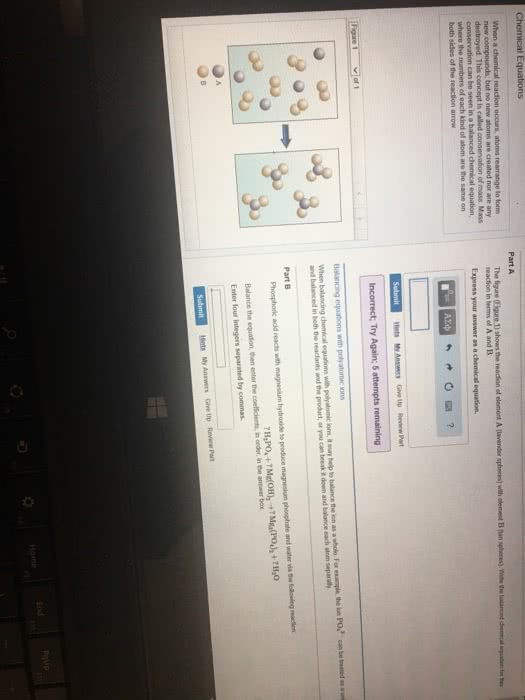
Labels for states of matter for reactants/products: (aq) for aqueous solutions, aka substances dissolved in water. Balanced chemical equation: when a chemical equation conforms to the law of conservation of matter. Chemical equations that are unbalanced (different number of atoms, of a certain element, on each side of the equation) need to be balanced by using coefficients. Coefficients = numbers that precede each formula and indicate how many of each formula must be involved in the reaction. When there is no coefficient in front of a reactant/product, it is assumed that the coefficient is 1 in that case. Stoichiometry = the study of the relationships between amounts of reactants and products in chemical reactions. Stoichiometric coefficients = the coefficients in a balanced chemical equation. It is conventional, but not always essential, to make the coefficients the smallest possible whole numbers. Write an unbalanced equation containing the correct formula of each reactant and.