CHEM 1112 Chapter Notes - Chapter 13: Solution, Miscibility, Strong Electrolyte
Document Summary
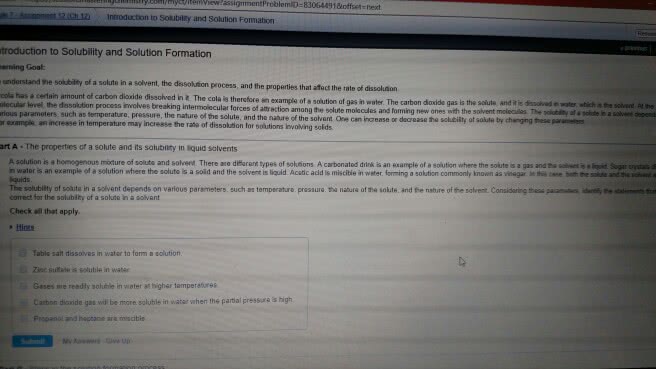
Solution is a homogeneous mixture of two or more substances: contains: solvent + solutes. Aqueous solutions solutions which water is the solvent. Saturated solution contains the maximum amount of solute that will dissolve in solvent at specific temperature. Unsaturated solution contains less solute than it has the capacity to dissolve. Supersaturated solution contains more dissolved solute than is present in a saturated solution: unstable. The majority of ionic salts become more soluble as the temperature increases because the water molecules will have more energy to move around and break the chemical bonds between the ionic compounds. Henry"s law states that the solubility of a gas in a liquid is proportional to the pressure of the gas over the solution. C = kp c: molar concentration (mol/l) of dissolved gas: p: pressure (atm) of the gas over the solution, k: is proportionality constant called the henry"s law constant (mol/l * atm)