CHEM 105bL Chapter 13: Chapter 13 - Chemical Kinetics
Document Summary
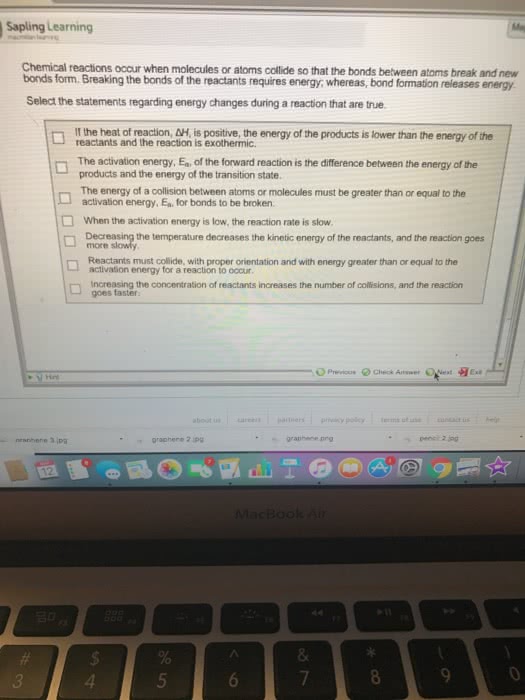
The rate law: the effect of concentration on the rxn. The rate law is used to express the relationship. To get from the reactants to the products, the molecule must go through a high-energy state called the activation complex or transition state. The exponential factor is a # between 0 and 1 that between the rate of the rxn and the concentration of the reactant. For (cid:1827) (cid:1868)(cid:1870)(cid:1867)(cid:1856)(cid:1873)(cid:1855)(cid:1872)(cid:1871), the rate law is (cid:1870)(cid:1853)(cid:1872)(cid:1857)=(cid:1863)[(cid:1827)](cid:3041) k is the rate constant and n is the rxn order. If n=0, then the rate is independent of the concentration of a. If n=1, then the rate is directly proportional to the concentration of a. If n=2, then the rate is proportional to the square of concentration of a. Zero order rxns occur when the amount of reactant available for the rxn is unaffected by changes in the overall quantity of reactant.