CHEM 112 Chapter Notes - Chapter 14: Reaction Rate, Stoichiometry, Reagent
Document Summary
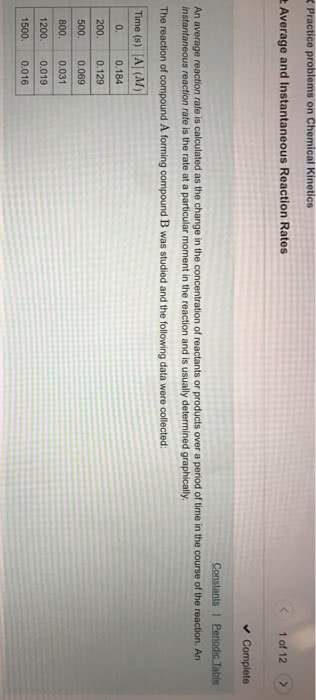
Reaction rate: speed at which a reaction progresses, as a ratio of change in concentration over change in time. Because a decreases over time, the change in its concentration is negative. Reaction rate is defined using the negative of the change in a reactant concentration over the change in time. Average reaction rate: obtained from the change in concentration of a reactant or product a defined time interval. Instantaneous rate: rate of a reaction at that point in time. Equal to the sole of a line tangent to the concentration-time curve at a given point in time. Initial rate: rate of the reaction at the beginning of the reaction. Reactant concentrations are easily measured at the start of a chemical reaction. When a is large, the slope of the curve is steep and thus the reaction is fast. When a is much smaller, the slope is smaller and the reaction slows.