CHEM 112 Chapter 3: Unit_3_Notes
Document Summary
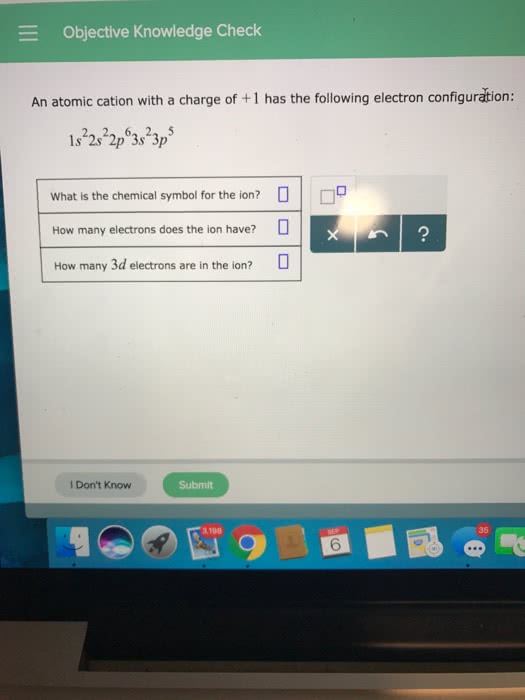
Unit 3 - chemical bonding and molecular structure. Valence electrons outer energy level electrons: electron-dot notation, an electron- configuration notation in which only the valence electrons of an atom of a particular element are shown, indicated by dots placed around the element"s symbol. Inner shell electrons are not shown: these are the electrons usually involved in the formation of covalent bonds. Ii: the octet rule ionic compounds. Ionic compounds tend to form so that each atom, by gaining, or losing electrons, has an octet of electrons in its highest occupied energy level. Formation of ions: electron configuration changes, cations lose their valence electrons to attain a noble-gas configuration, formation of a sodium ion, anions gain electrons to complete their valence shell noble-gas configuration, formation of chloride ion. Oppositely charged ions come together in a ratio that produces a net charge = 0.