CHEM 14C Chapter Notes - Chapter 1: Uncertainty Principle, Atomic Orbital, Electronegativity
Document Summary
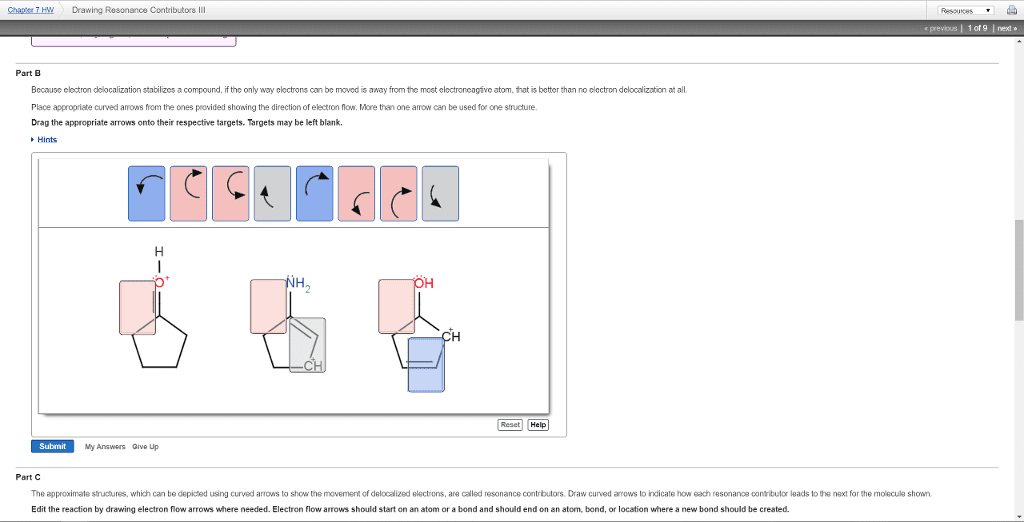
4. never exceed octet count for elements in second row. 5. not all resonance forms are equivalent sometimes one contributes more than others. 3. structures with max octets are most important charges should be preferentially located on atoms with compatible electronegativity structures with less separation of opposite charges are more important resonance contributors than those with more charge separation. A. separating opposite charges requires energy neutral structures are better than dipolar ones coulomb"s law a. in some cases to draw octet lewis structures, charge separation is necessary so 1 >> 3 rules: draw lewis structures molecular skeleton. # valence electrons how many bonds do we need (total - valence) / 2 connect determine formal charges evaluate resonance forms. 2s = sphere -- larger than 1s, higher energy, farther from positive nucleus. 2px, 2py, 2pz = two lobes ( gure 8) solutions of equal energy of this type = degenerate assigning electrons to orbitals.