CHEM 14A Chapter Notes - Chapter 2F.1: Triple Bond, Sigma Bond, Unpaired Electron
Document Summary
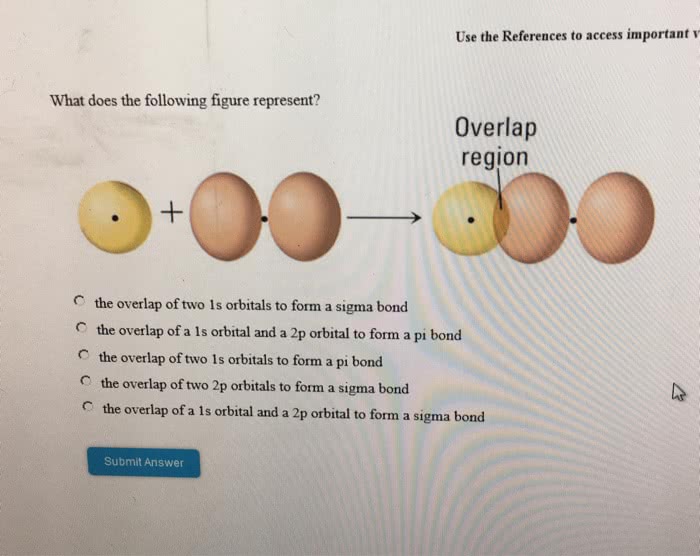
Valence-bond theory (vb theory): the description of covalent bonding in terms of atomic bonding. Takes the wave nature of electrons into account. Valence-bond theory adopts the view that, as two h atoms come together, their 1s-electrons pair and two atomic orbitals merge together. Sigma-bond: cylindrically symmetrical with no nodal planes containing the internuclear axis. Overlap of orbitals: the merging of the two atomic orbitals. The greater the extent of orbital overlap, the stronger is the bond. There"s a single electron in each of the 2p-orbitals on each atom. However, only one of the three orbitals on each atom can overlap end-to-end to form a sigma bond. Two of the 2p-orbitals on each atom are perpendicular to the internuclear axis, each containing an unpaired electron. Pi bond: a single nodal plane containing the internuclear axis. Double bond: sigma bond and one pi bond. Triple bond: 1 sigma bond and two pi bonds.