CHEM 14A Chapter Notes - Chapter 2C.1: Antioxidant, Methyl Radical, Unpaired Electron
Document Summary
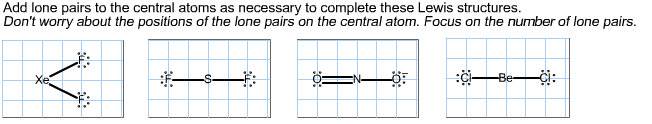
The octet rule accounts for the valences of many of the elements and structures of. Topic 2c beyond the octet rule many compounds. A molecule might have an odd number of electrons, so octet formation is numerically impossible. Atoms of certain elements might be able to accommodate more than eight electrons in their valence shells. An atom might form compounds with incomplete octets. Some species have an odd number of valence electrons. Radicals: species having electrons with unpaired spins. The single unpaired of electron for ch3 is indicated by a dot ( ch3) Radicals are of crucial importance for the chemical reactions that take place in the upper atmosphere. Contribute to the formation and decomposition of ozone. Antioxidant: an additive that delays damages from radicals. One unpaired electron is on one carbon atom of the chain and the second is on another carbon atom several bonds away.