CHEM 1C Chapter Notes - Chapter 17: Lewis Acids And Bases, Rice Chart, Ion
Document Summary
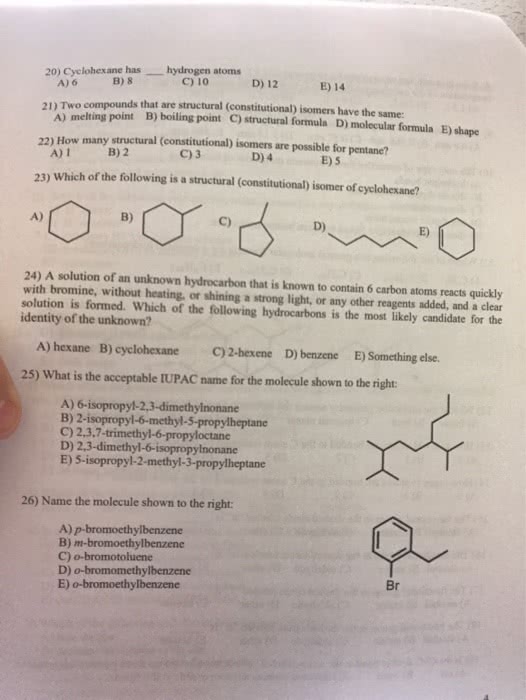
Acids have sour taste, ability to dissolve many metals, turn blue litmus paper red, neutralize bases. Carboxylic acid = contains the following grouping of atoms. Bases have a bitter taste, a slipper feel, turn red litmus paper blue, neutralize acids. Alkaloids = organic bases found in plants that are often poisonous. 17. 3 definitions of acids and bases: the arrhenius definition. Acid = a substance that produces h+ ions in an aqueous solution. Base = a substance that produces oh- ions in aqueous solution. Strong base = one that completely dissociates in solution (analogous to strong acid) Weak base = analogous to a weak acid: the bronsted-lowry definition. Br nsted-lowry definition = focuses on transfer of h+ ions in an acid-base reaction. Conjugate acid-base pair = two substances related to each other by the transfer of a proton\ Conjugate acid = is any base to which a proton has been added.