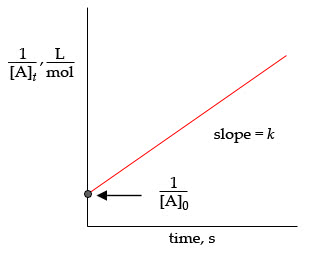
For a second-order reaction,
[A]?products, the rate of the reaction is given as
rate= k[A]2, where
k is the rate constant and
[A] is the concentration of reactant
A. The integrated rate law for second-order reactions is
1[A]t=kt+1[A]0, where
[A]t is the concentration of reactant
A at time
t,
k is the rate constant, and
[A]0 is the initial concentration of reactant
A. This equation is of the type
y=mx+b. Therefore, the plot of
1[A]t versus time is always a straight line with a slope
k and a
y intercept
1[A]0.
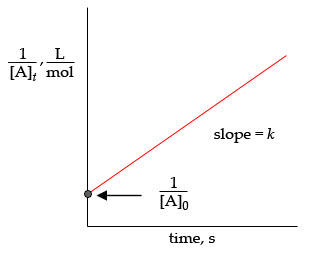
Part C
For a second-order reaction, the rate constant k is the slope of the graph of 1[A] versus t. Based on this information and the data given, calculate the rate constant k for the reaction.
Express your answer in M?1?min?1 to three significant figures.
For a second-order reaction, [A]?products, the rate of the reaction is given as rate= k[A]2, where k is the rate constant and [A] is the concentration of reactant A. The integrated rate law for second-order reactions is 1[A]t=kt+1[A]0, where [A]t is the concentration of reactant A at time t, k is the rate constant, and [A]0 is the initial concentration of reactant A. This equation is of the type y=mx+b. Therefore, the plot of 1[A]t versus time is always a straight line with a slope k and a y intercept For a second-order reaction, the rate constant k is the slope of the graph of 1[A] versus t. Based on this information and the data given, calculate the rate constant k for the reaction. Express your answer in M?1?min?1 to three significant figures.