CHEM 200 Chapter Notes - Chapter 7: Electromagnetic Spectrum, Ultraviolet, Radiant Energy
Document Summary
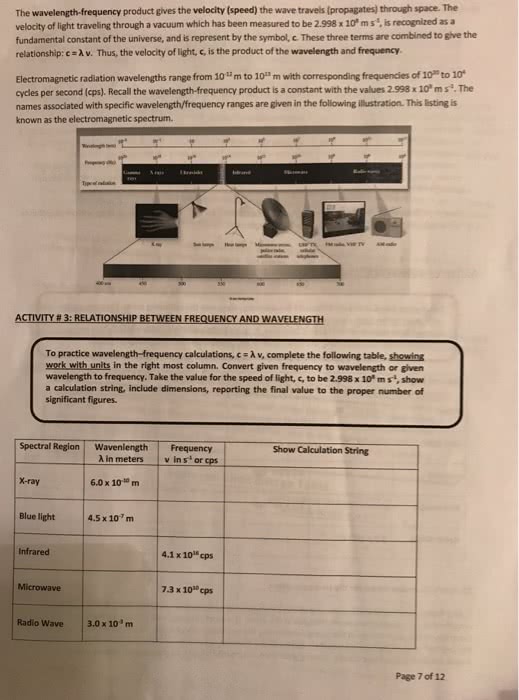
Chapter 7 quantum theory and atomic structure. Electromagnetic radiation visible light, x-rays, and microwaves; consists of energy propagated by electric and magnetic fields that increase and decrease in intensity as they move through space. Visible light represents a small region of the electromagnetic spectrum. Electromagnetic spectrum the continuum of wavelengths of radiant energy. All waves in the spectrum travel at the same speed through a vacuum but differ in frequency and wavelength. The spectrum is a continuum of radiant energy, so each region meets the next. Infrared (ir) the region of the electromagnetic spectrum between the microwave and visible regions. We perceive different wavelengths (or frequencies) of visible light as colors, from red to violet. Polychromatic: light of many wavelengths; example: white light. The region adjacent to visible light on the short-wavelength end consists of ultraviolet radiation between the visible and the x-ray regions. Ultraviolet (uv) radiation in the region of the electromagnetic spectrum.