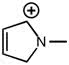
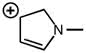
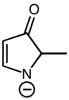
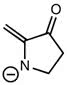
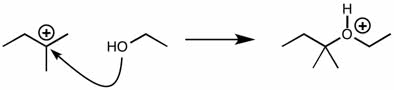
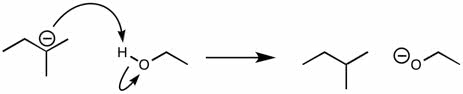
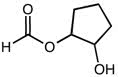
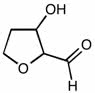
CHEM 1210 Chapter Notes - Chapter 11: Lewis Structure, Linear Combination, Molecular Orbital Diagram

37
CHEM 1210 Full Course Notes
Verified Note
37 documents
Document Summary
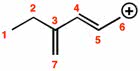
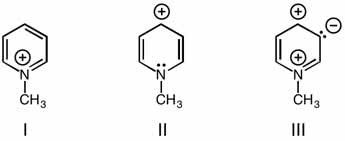
11-2 introduction to the valence-bond method: when two h atoms approach each other, the region of high electron probability interpenetrate. 11-3 hybridization of atomic orbitals: sp2 hybrid orbitals. 11-5 molecular orbital theory: lewis structures, vsepr theory, and the valence-bond method do not provide an explanation of the electronic spectra of molecules, why oxygen is paramagnetic or why h2 species. It enters the s1s orbital, a bonding molecular orbital. Using equation (11. 1), we see that the bond order is 11 - 02>2 = 12. This is equivalent to a one-electron, or half, bond, a bond type that is not easily described by the lewis theory: he2 two electrons are in the orbital, and two are in the . The bond order is 12 - 22>2 = 0. The greater the electron density the higher the bond order: bonding in the molecule h2so4 and anion so4.