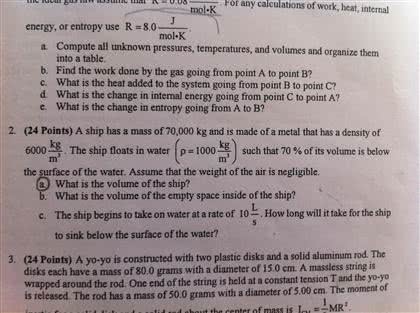PHYS 2001 Chapter : Laws Of Thermodynamics
Document Summary
Laws of thermodynamics (a two-page primer) int dq de int dw. Statement: when two objects have equal temperatures and are in thermal contact with each other they must be in thermal equilibrium. (and, therefore, they must have equal internal energies. ) In other words: every object has a property called temperature. " when two objects are in thermal equilibrium with each other, they have equal temperatures. The internal energy of a system increases if energy is added via heat and decreases if energy is lost via work done by the system. In other words: you can"t get something for nothing . Definition: a heat engine is any device that can change thermal energy into useful mechanical work (examples: steam engines, internal combustion engines, heat pumps, refrigerators). Statement 1: heat flows naturally from a hot object to a cold object; heat will not flow spontaneously from a cold object to a hot object. (clausius" statement of the 2nd law. )