CHEM 1202 Chapter : Chapter 14 Exam Review Notes
Document Summary
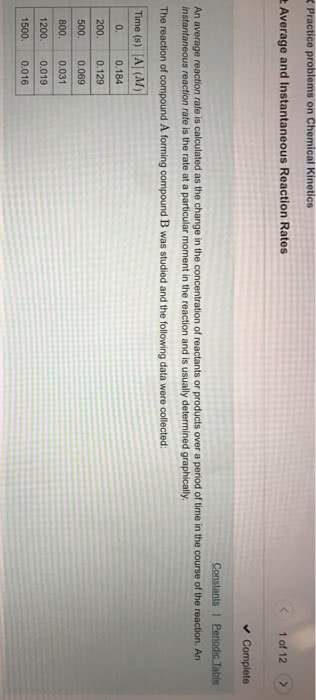
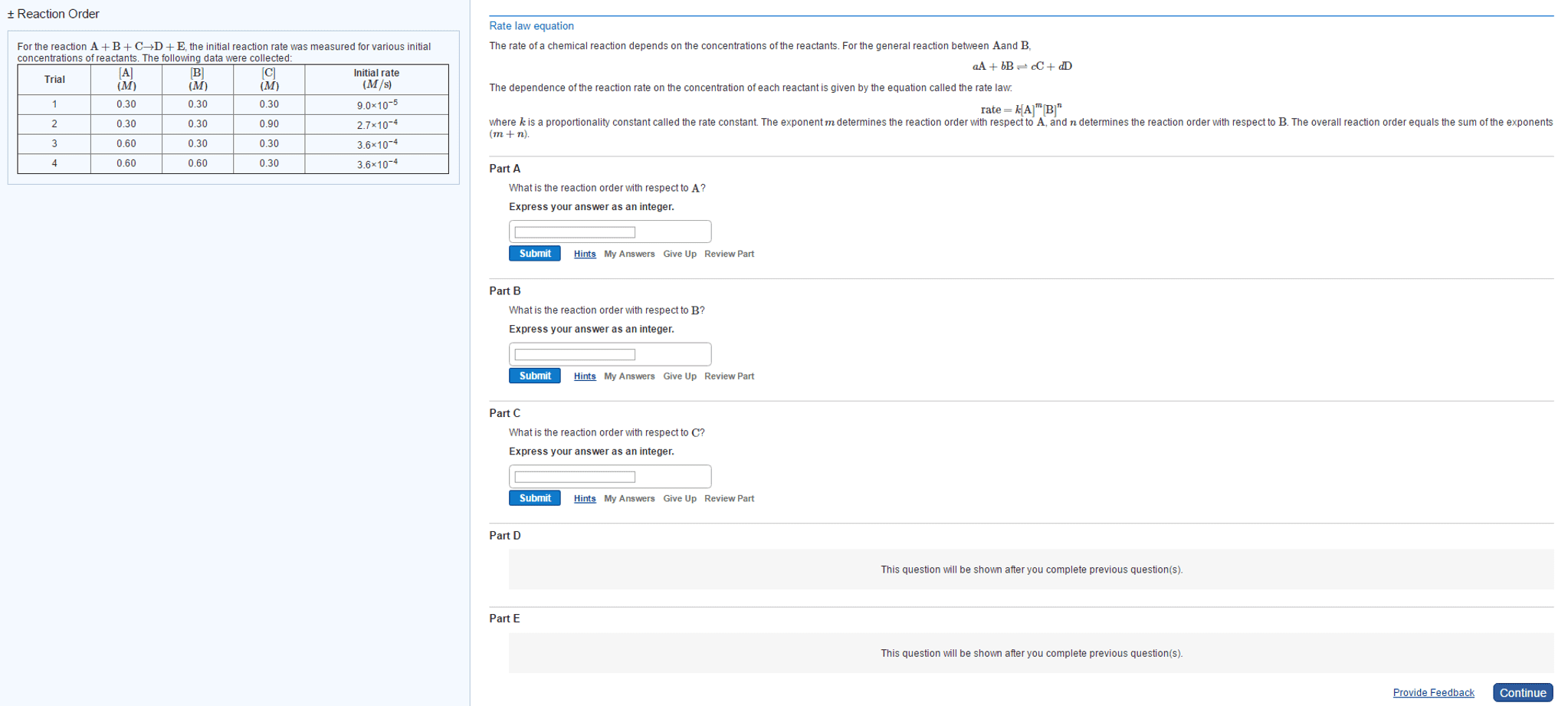
Chapter 14 kinetics: average rate of reaction, a rate of reaction calculated over an interval of time is called the average rate of reaction, rates of reactions are always positive. Instantaneous rate: rate of reaction calculated at a specific instant in time is called the instantaneous rate of reaction, using chemical equations to compare rates of reaction, rates of reaction are related by the stoichiometry. The average rate of reaction for reactant a is avg rate=- a/ t: rate law. This general rate law describes how the rate of reaction depends on the concentration of reactants. k is a rate constant that depends on the reaction, temperature, and the existence of a catalyst: rate= k[reactant a]m[reactant b]n. The reaction order refers to the exponents, m, and n. the overall order is the sum of the exponents (m+n): note: the exponents are determined experimentally, they are not always necessarily the exponent numbers.