CHEM 1202 Chapter : Exam 1 Outline Ch 15 16 And 17 4
Document Summary
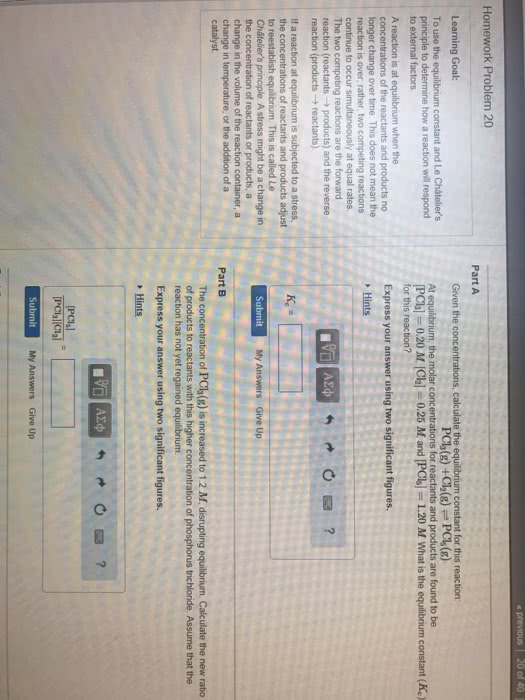
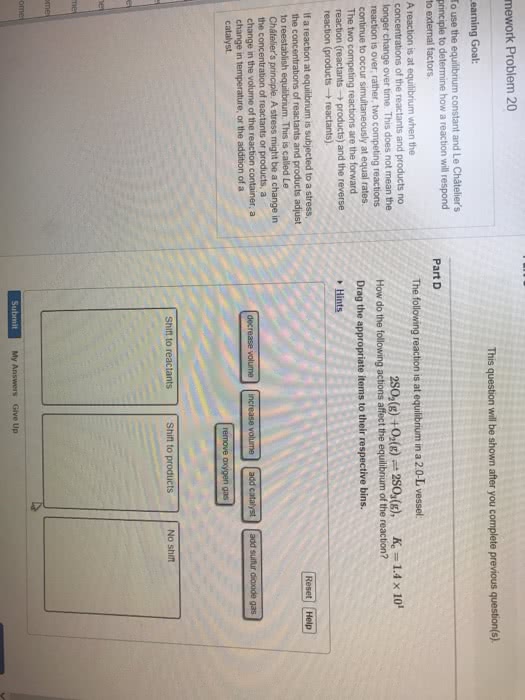
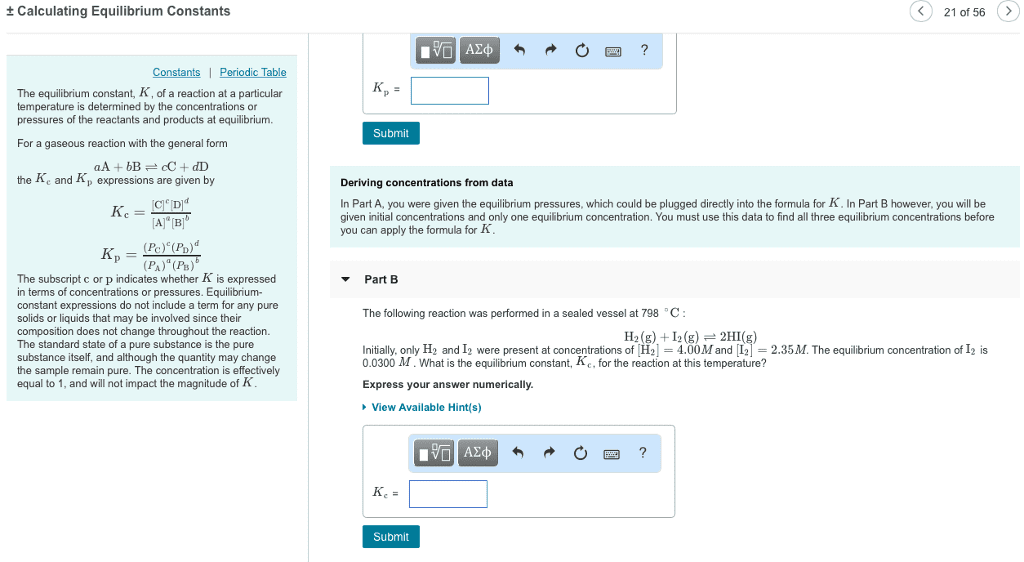
If given k, determine if the products or reactants dominate at equilibrium. (cid:120) a negative power of 10 indicates that it is a number less than 1. Emphasize that k does not change. (cid:120) effects of volume and pressure changes for a gaseous equilibrium mixture. Chapter 16 outline acid-base equilibria (cid:120) (cid:120) Identify an acid/base using the arrhenius definition (cid:120) write the reaction when an acid/base is dissolved in water. Emphasize that only equilibrium concentrations can be used in the equilibrium-constant expression. If a solution is basic, then [oh-] > [h+]. M, because two oh- ions are generated for every one ba(oh)2 molecule). (cid:120) weak acids only partially ionize in solution and will exist in equilibrium. (cid:120) ka is the acid-dissociation constant. They are not needed if the difference between ka1 and ka2 is 103 or greater. (cid:120) weak bases only partially accept h+ in solution and will exist in equilibrium. (cid:120) kb is the base-dissociation constant.