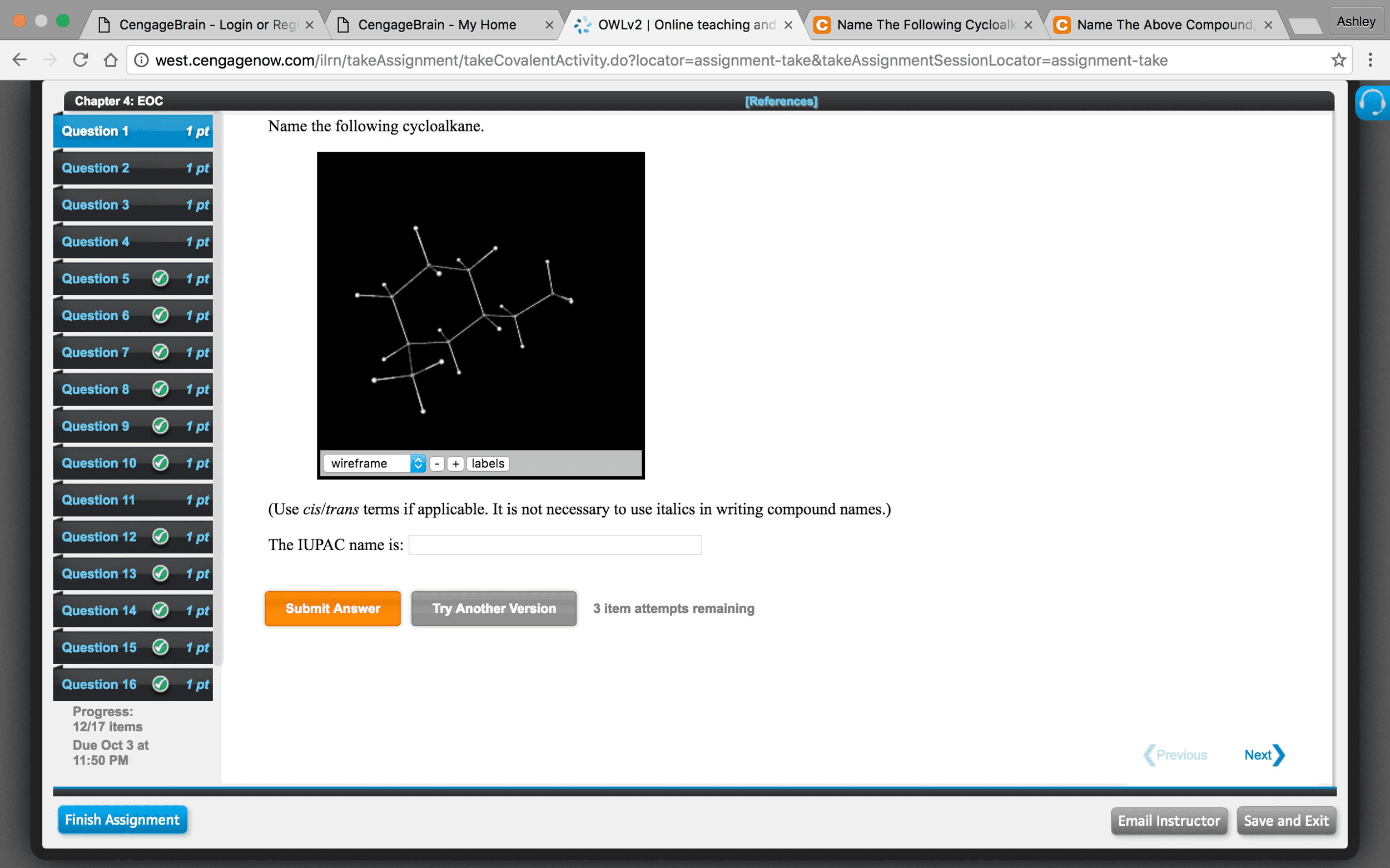
17 Data Analysis Molecular Geometry,I Using molecular models, construct the following molecules/polyatomic ions, and write their Lewis formulas. Record the following information for each in your laboratory notebook. a) Molecule or ion (Formula and IUPAC name) b) Lewis structure c) Draw any resonance structures if applicable d) # of bonding regions (double and triple bonds count as a single region) e) # of nonbonding pairs f) Hybridization of the central atom g) VSEPR designation h) i) Molecular geometry of the molecule/polyatomic ion, Polarity of the molecule/ion i) 3-D sketch Your table in your notebook should be neat and organized to include all of the following information for each of the 16 molecules/ions: Molecule or Polyatomic lon Lewis Structure (include any resonance structures) Data Sketch of the 3-D Bonding regions # of bonding regions # of non-bonding pairs Hybridization VSEPR formula Molecular geometry Polar or nonpolar 1. CF,CI 3. NO 4. HNO 5. PHs 6. HCN 7. PO. 8. Gal 9. BrF 10. SF4 11. XeOF 12. AsF 13. IF 14. so, 15. AsFs 16. XeF2 17. Dimethyl sulfide; for fun facts see http://www.chm.bris.ac.uk/motm/dimethylsulphide/dmsh.htm Molecular Geometry
Show transcribed image text 17 Data Analysis Molecular Geometry,I Using molecular models, construct the following molecules/polyatomic ions, and write their Lewis formulas. Record the following information for each in your laboratory notebook. a) Molecule or ion (Formula and IUPAC name) b) Lewis structure c) Draw any resonance structures if applicable d) # of bonding regions (double and triple bonds count as a single region) e) # of nonbonding pairs f) Hybridization of the central atom g) VSEPR designation h) i) Molecular geometry of the molecule/polyatomic ion, Polarity of the molecule/ion i) 3-D sketch Your table in your notebook should be neat and organized to include all of the following information for each of the 16 molecules/ions: Molecule or Polyatomic lon Lewis Structure (include any resonance structures) Data Sketch of the 3-D Bonding regions # of bonding regions # of non-bonding pairs Hybridization VSEPR formula Molecular geometry Polar or nonpolar 1. CF,CI 3. NO 4. HNO 5. PHs 6. HCN 7. PO. 8. Gal 9. BrF 10. SF4 11. XeOF 12. AsF 13. IF 14. so, 15. AsFs 16. XeF2 17. Dimethyl sulfide; for fun facts see http://www.chm.bris.ac.uk/motm/dimethylsulphide/dmsh.htm Molecular Geometry