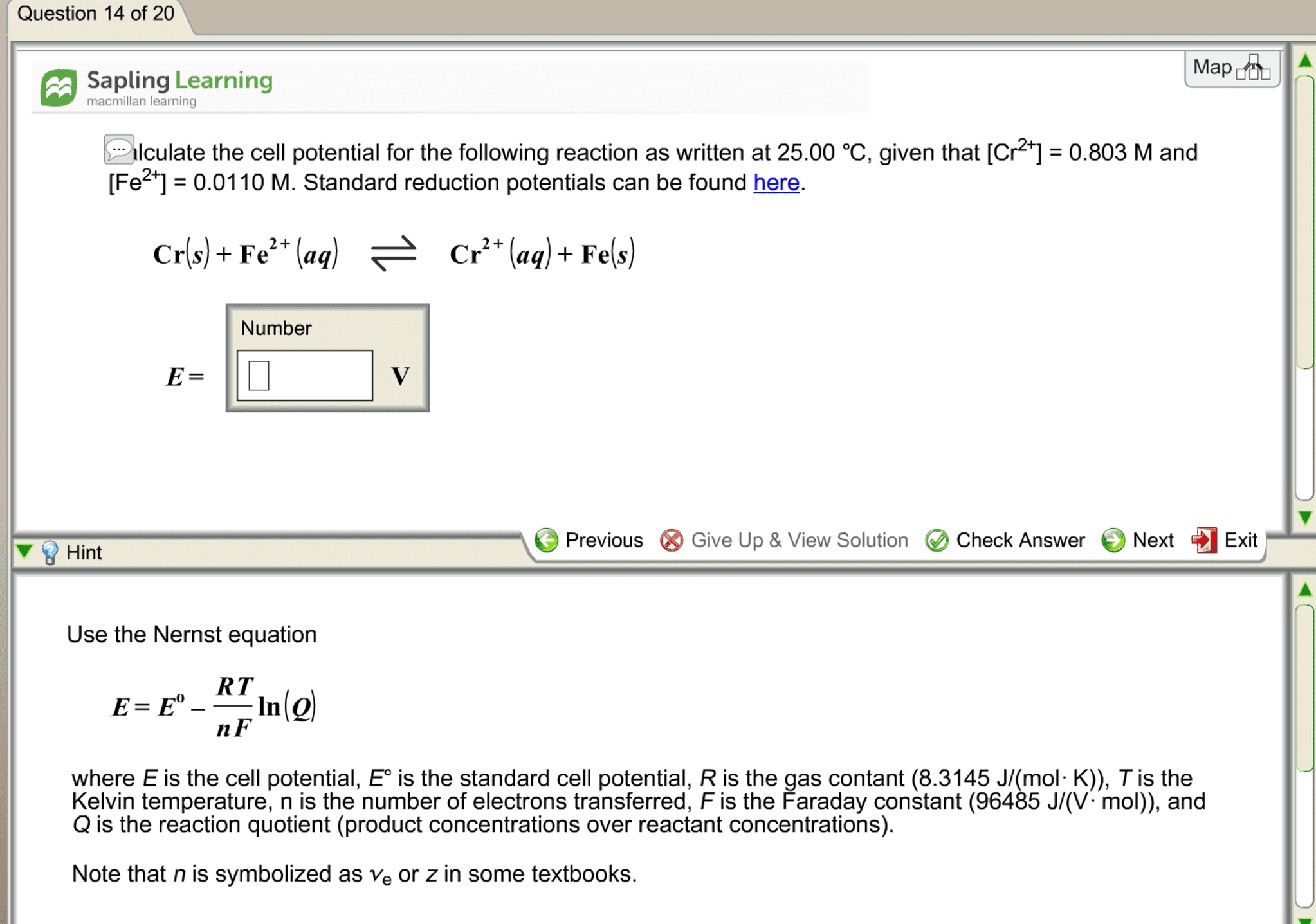
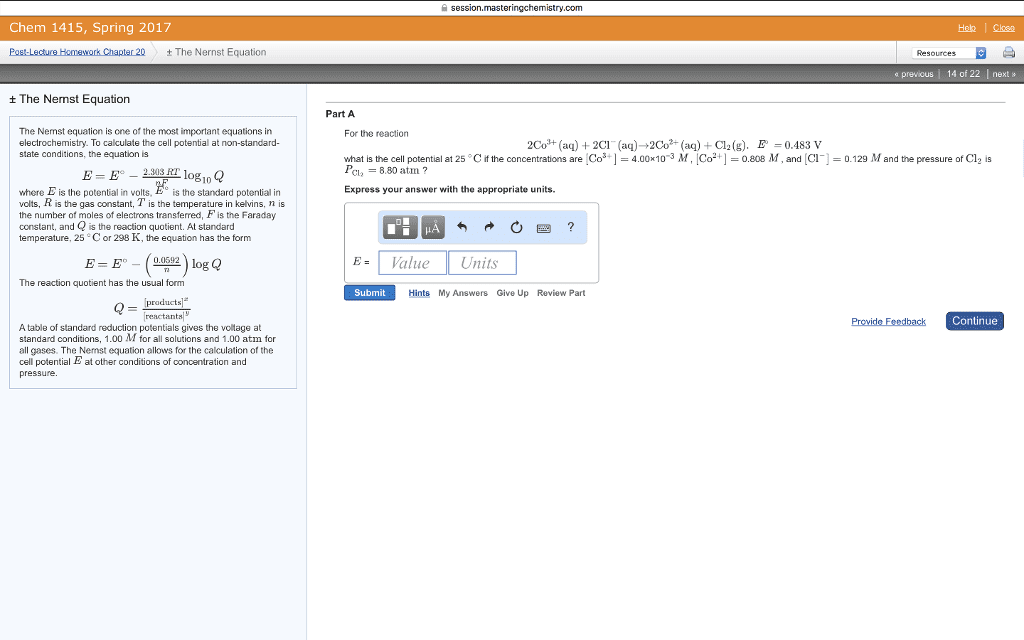
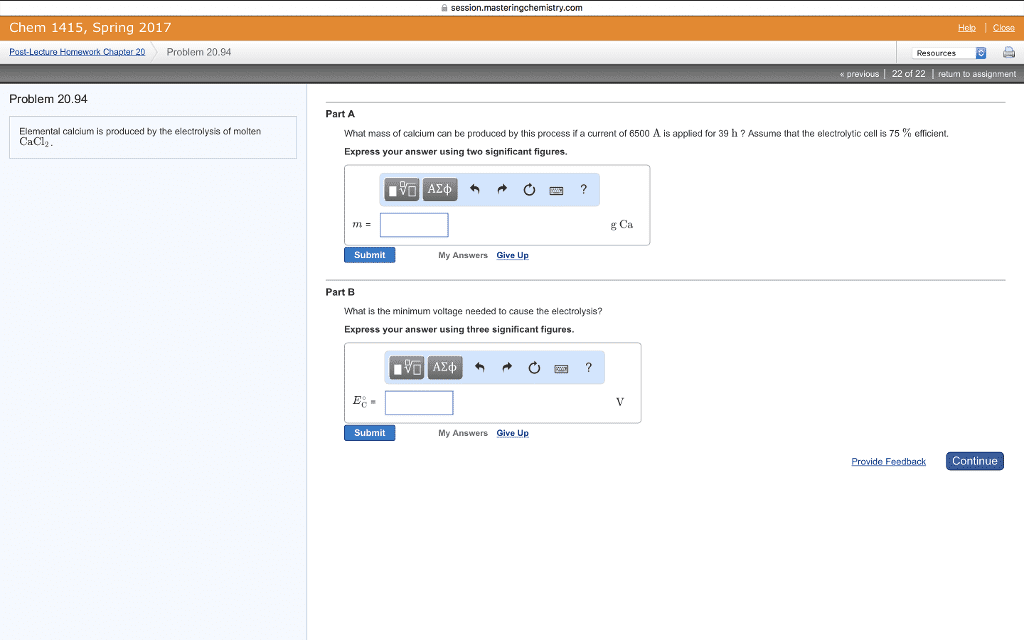
CHEM 1A Chapter Notes - Chapter 3: Nernst Equation, Electrochemical Cell, Concentration Cell
Document Summary
Nernst equation: as reactants are consumed in a working electrochemical cell decreases until finally it reaches zero, dead cell battery = cell reaction has reached equilibrium. , the cell potential also expressing the potential of an electrochemical cell in terms of concentrations of the reagents . In q at 298. 15k , rt if = 0. 025693 v, O - 025693 v yn o, h r using logarithms , E cell = eceii - rt!? " y rfi = 0. 025693 v log q = eai, Cag , 11 cult cag ) ) cuts) Cu"cag ) -12ms) 2h"- lag ) 1- cues ) 001 concentration cell : a galvanic cell in which the electrodes have the same composition but are at different concentrations. No driving force for change when the two concentrations are the same. In q example : a concentration cell having two ag -11 ag electrodes.