Chemistry 2213A/B Chapter Notes - Chapter 6: Polarimeter, Chemical Formula, Cyclopentane
Document Summary
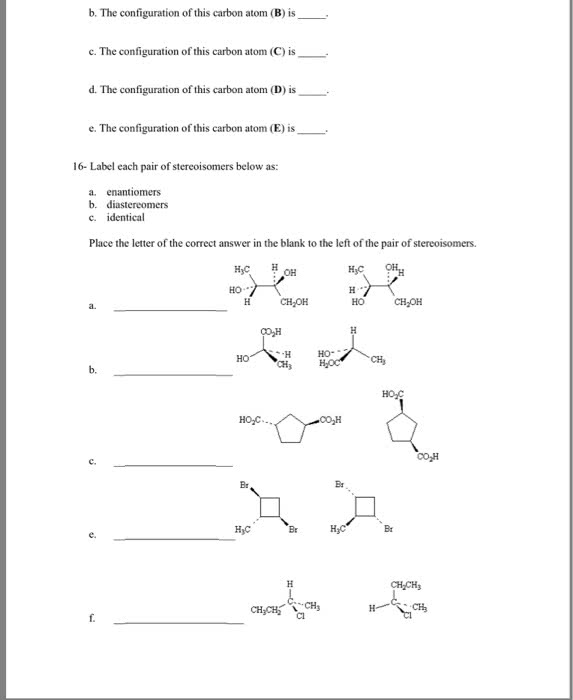
Chapter 6: chirality the handedness of molecules. Stereoisomers: same molecular formula and same connectivity of atoms but different three dimensional orientation of atoms. Stereoisomers: same formula and same connectivity but not mirror images: enantiomers: non superimposable mirror images, diastereomers: not mirror images. 6. 3 how do we designate the configuration of a stereocenter: 6. 4 what is the 2n rule: n stereocenters warrants 2n stereoisomers. Enantiomers and diastereomers: enantiomers: stereoisomers that are non superimposable mirror images, diasteriomers: stereoisomers that are not mirror images of each other. Look at the compound confirm same chemicall formula, find r/s configurations, compare the configurations if they are completely opposite then they are enantiomers, otherwise diasteriomers. 6. 5 how do we describe the chirality of cyclic molecules with two structures: Disubstituted derivatives of cyclopentane: the same rule for meso applies to rings if it is symmetrical then there is a loss of chirality. If we have cis/cis with the same side chains then the compound is meso.