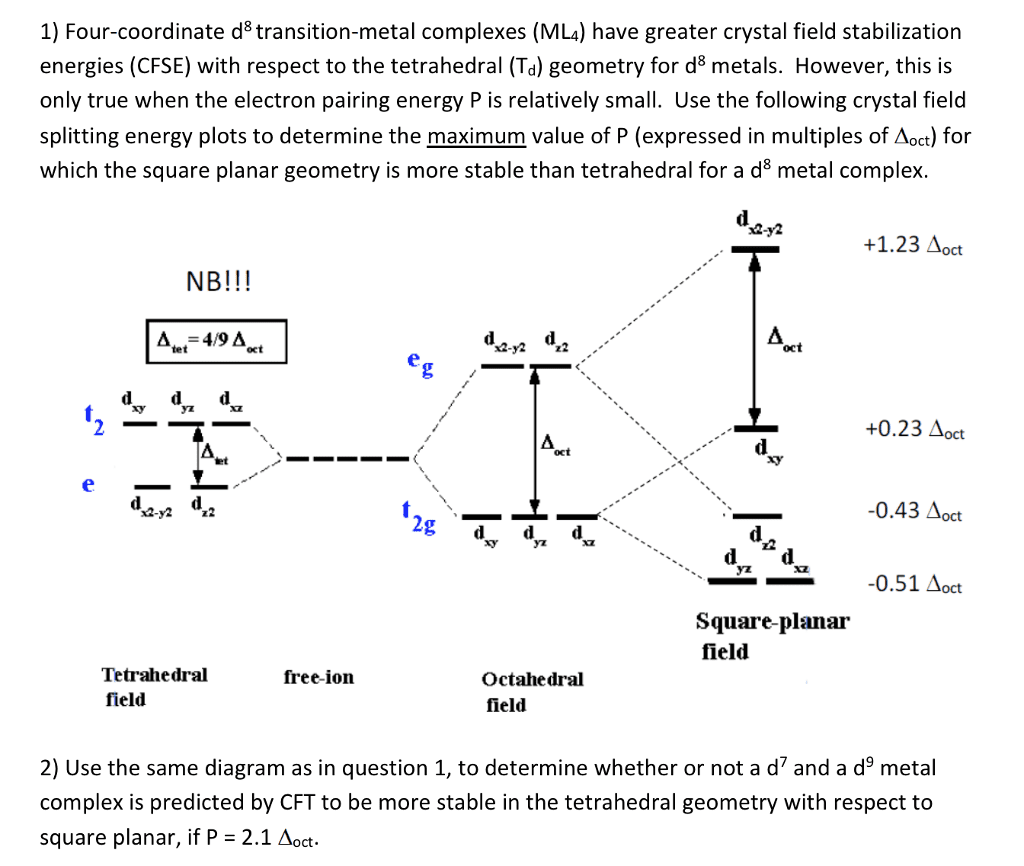
The final amino copper(ll) complex you've made has a 5-coordinate, square pyramidal structure, with a ligand in the apical site. We can use crystal field theory to describe the bonding between a copper atom and the nitrogen and oxygen atoms in the complex. This assumes that the bonding is electrostatic, and upon the number of d electrons at the metal centre. that the magnetic properties of the complex are only dependent The diagram below shows that as the nitrogen and oxygen atoms attach to the copper atom, the energies of the metal d-orbitals change. Electrons from the metal will fill these orbitals, starting from the lowest energy first. The oxidation state of the metal in the complex determines the number of electrons in this diagram. 0 Use the labelled crystal field energy diagram provided below to show the correct distribution of metal electrons in this amino complex. Energy Square pytamidal (i) Comment below, with your reasoning, on whether the copper to apical ligand distance will be longer or shorter than expected. You should assume the metal-apical ligand bond occurs along the z axis Do you expect this complex to be diamagnetic or paramagnetic, explain your answer below i)
Show transcribed image text The final amino copper(ll) complex you've made has a 5-coordinate, square pyramidal structure, with a ligand in the apical site. We can use crystal field theory to describe the bonding between a copper atom and the nitrogen and oxygen atoms in the complex. This assumes that the bonding is electrostatic, and upon the number of d electrons at the metal centre. that the magnetic properties of the complex are only dependent The diagram below shows that as the nitrogen and oxygen atoms attach to the copper atom, the energies of the metal d-orbitals change. Electrons from the metal will fill these orbitals, starting from the lowest energy first. The oxidation state of the metal in the complex determines the number of electrons in this diagram. 0 Use the labelled crystal field energy diagram provided below to show the correct distribution of metal electrons in this amino complex. Energy Square pytamidal (i) Comment below, with your reasoning, on whether the copper to apical ligand distance will be longer or shorter than expected. You should assume the metal-apical ligand bond occurs along the z axis Do you expect this complex to be diamagnetic or paramagnetic, explain your answer below i)