CHM120H5 Chapter Notes - Chapter 10: Paramagnetism, Biomolecule, Fluorine
Document Summary
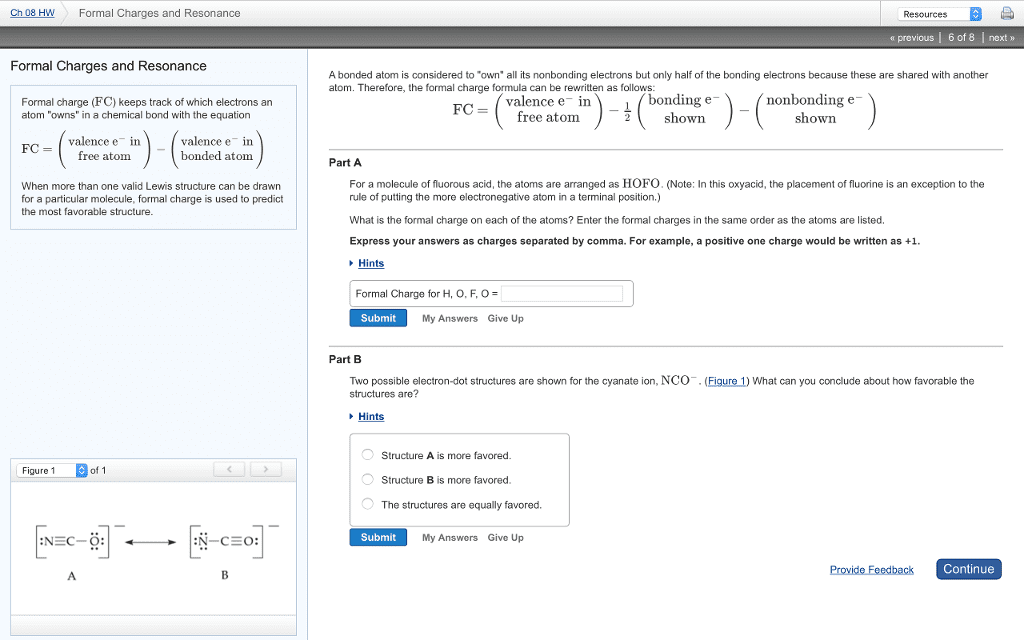
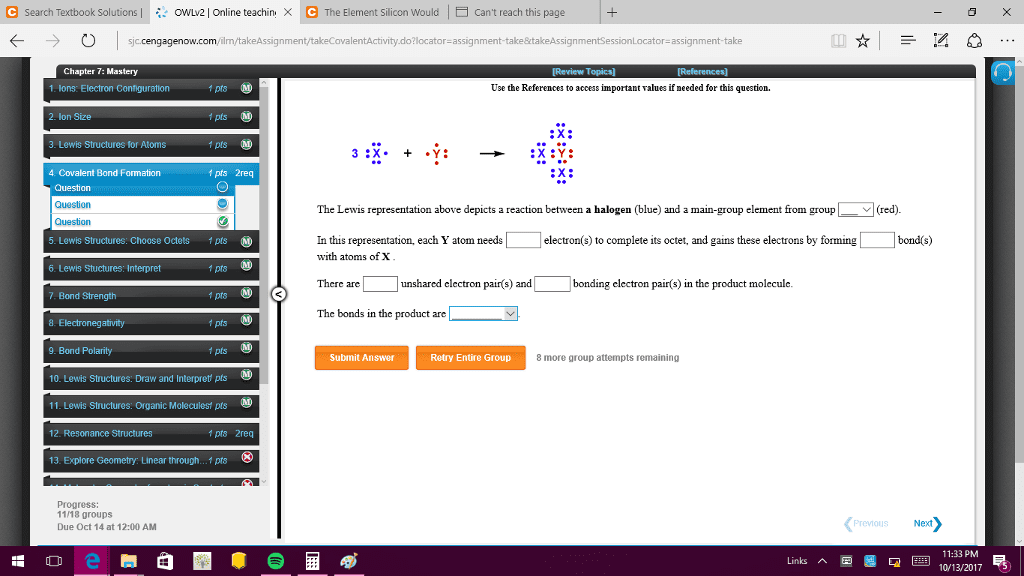
Chapter 10: the shapes of molecules: 1: depicting molecules and ions with lewis structures. Shows electron dot symbols for the atoms, and bonding pairs as line sand the lone pairs that fill each atom"s outer level (valence shell as pairs of dots) Applying the octet rule to write lewis structures: resonance: delocalized electron-pair bonding. In a single, double or triple bond each electron pair is localized b/w the bonded atoms. In a resonance hybrid, two of the electron pairs are delocalized: their density is spread over a few adjacent atoms. Partial bonding, as in resonance structures, often leads to fractional bond orders: formal charge: selecting the more important resonance structure. One way to select the more important resonance form is by determining each atom"s formal charge, the charge it would have if the bonding electrons were shared equally. Smaller formal charges(positive or negative) are preferable to large ones. The same nonzero formal charges on adjacent atoms are not preferred.