CHEM 313 Chapter Notes -Lewis Structure, Chch-Dt, Bayerischer Rundfunk
Document Summary
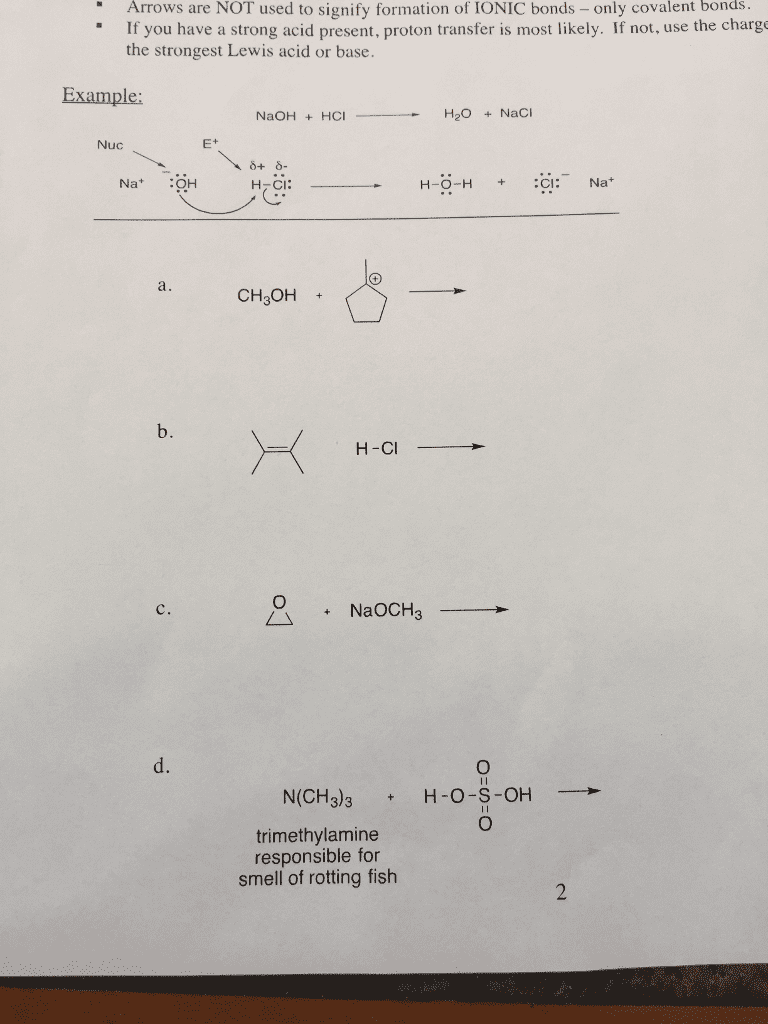
Select the more electronegative atom in each of the following pairs: bonding and electronics (c) o or s (e) s or p (a) f or i (b) n or c (d) n or o (f) n or p. Characterize the following bonds as ionic or covalent: Determine whether the atoms circled below have a full octet: Provide the lewis dot structures for the following compounds. (a) nh3 (b) nh4 (c) h2o (d) h3o+ (e) cl : 1. Determine the hybridization of the orbital of the circled lone pairs: Determine the hybridization of each of the indicated atoms. (a) carbon in ccl4 (c) carbon in hcn (e) carbon in co2 (b) nitrogen in nh3 (d) nitrogen in hcn. The compound with the structure below may be biologically active against arthritis. Determine the formal charge of each of the indicated atoms (not all lone pairs are shown). (a) oxygen in. Oh (b) nitrogen in (c) boron in (d) oxygen in.