CHEM 313 Chapter Notes -Tautomer, Ammonium Chloride, Phosphoramidate
Document Summary
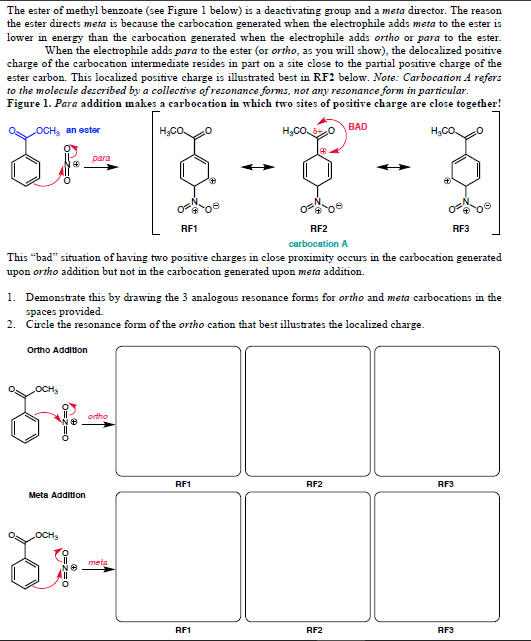
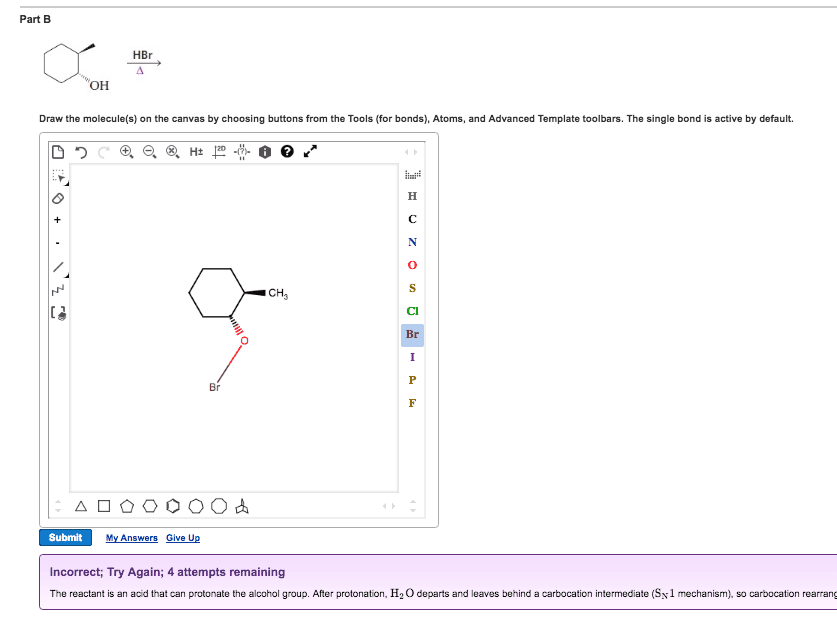
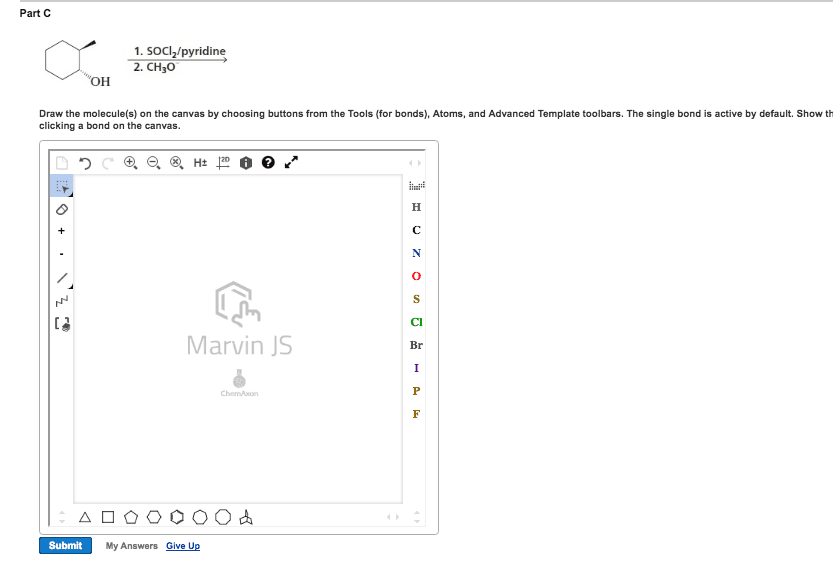
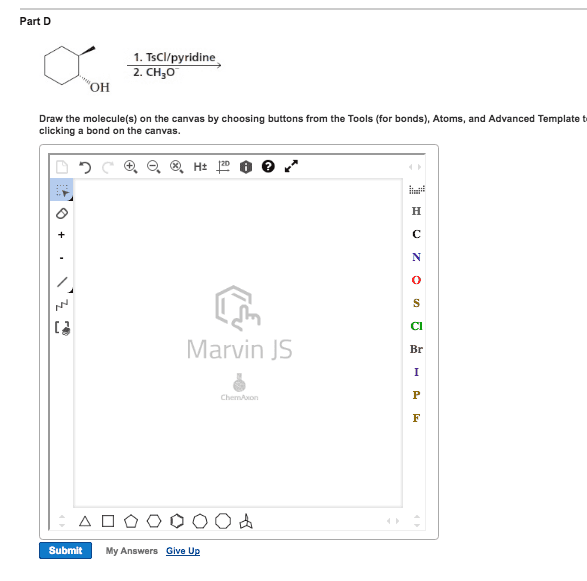
Chem 313 problem set #7 2014, answers: answer: Ome is an edg and stabilizes the carbocation through resonance: answers: No2 is an ewg and therefore destabilizes the carbocation: answer, answer, answer: compound b shows two products due to the tetrahedral nature of phosphorous. Upon removal of the cyanoethyl group, the chirality is lost (product c): answer, answer: the linker (4,4"-dimethoxytrityl) is acid labile. Resins 5 and 3 are not a good choice because the. Dna will be link by an ether which is difficult to cleave under the protocol seen in class. During the synthesis of the amide bond, an extra equivalent of base will be needed to deprotonated the ammonium yielding a free secondary amine ready to be acylated. 2013, 78, 8417: as illustrated in the proposed reaction path for 23, protonation of the inactive monoionic phosphoramidate 46 (p o /p nr2) generates the neutral phosphoramidate 47a (p oh/p nr2) and its zwitterionic tautomer 47b (p o /p nh+r2).