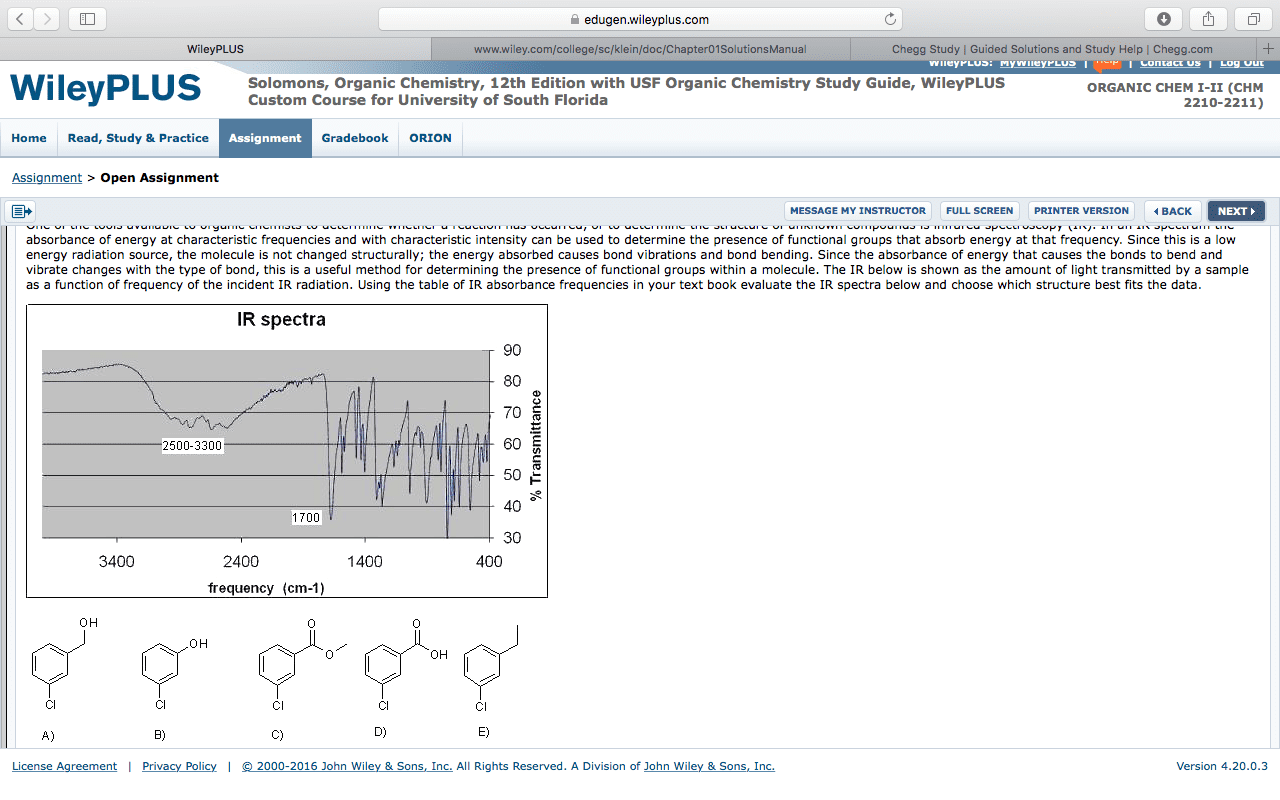
CHEM267 Chapter Notes - Chapter 15: Infrared Spectroscopy, Wavenumber, Saturated And Unsaturated Compounds
Document Summary
Ir radiation causes vibrational excitation of the bonds in a molecule. Different kinds of vibrational excitation; bonds can: stretch (like a spring, bend. For every bond in a molecule, the energy gap between vibrational states is dependent on the nature of the bond (ex. the energy gap for a c-h bond is much larger than for a c-o bond) Each type of bond will absorb a characteristic frequency: by irradiating the compound with al frequencies of ir radition, you can determine which types of bonds are present in a molecule by detecting which frequencies were absorbed. A sample is irradiated with frequencies of ir radiation, and the frequencies that pass through (those that are not absorbed by the sample) are detected. The general shape of an ir absorbance spectrum. Wavenumber is proportional to frequency; larger wavenumber = higher energy left side of plot (@ 4000) = higher energy radiation; right side (@ 400) = lower energy radiation.