CHEM103 Chapter 10: Ch.10 Chemical Bonding

128
CHEM103 Full Course Notes
Verified Note
128 documents
Document Summary
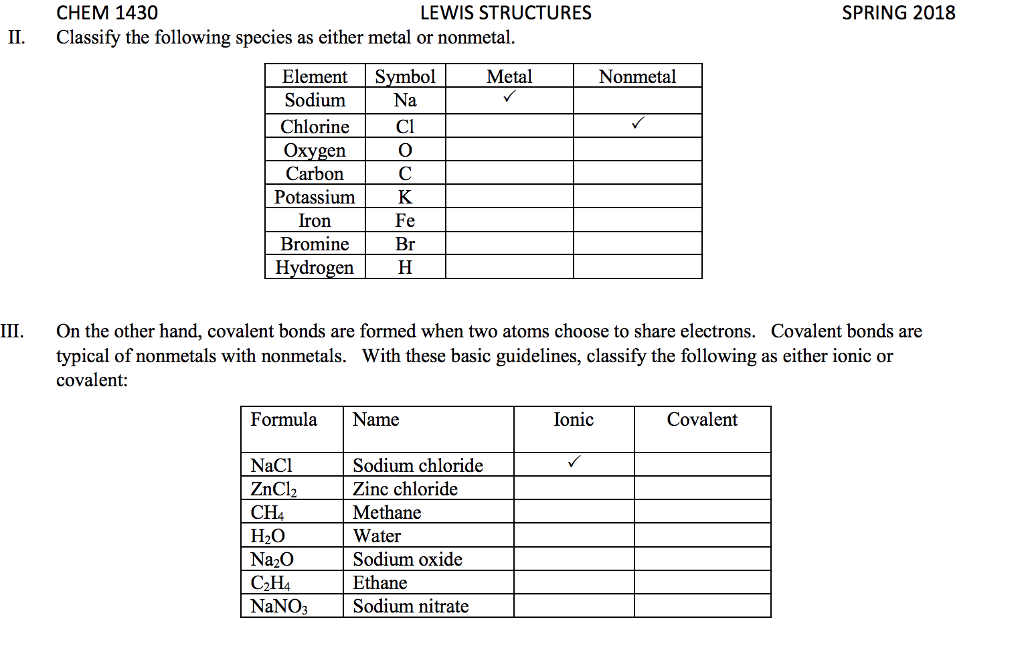
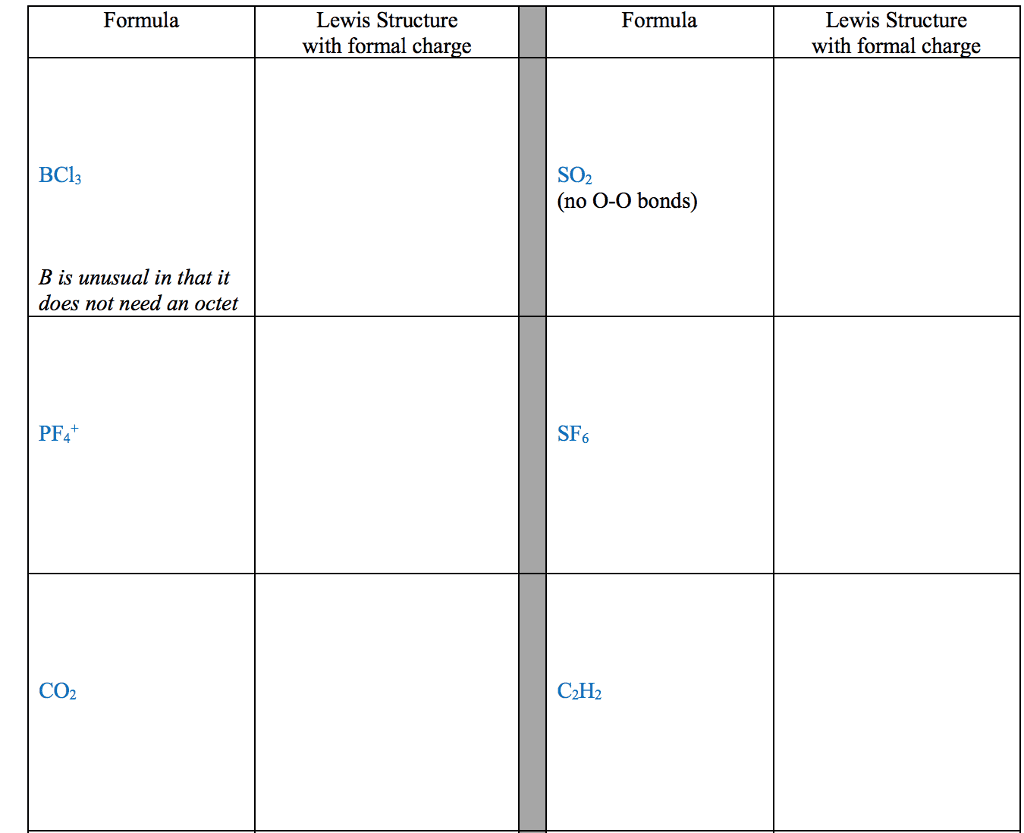
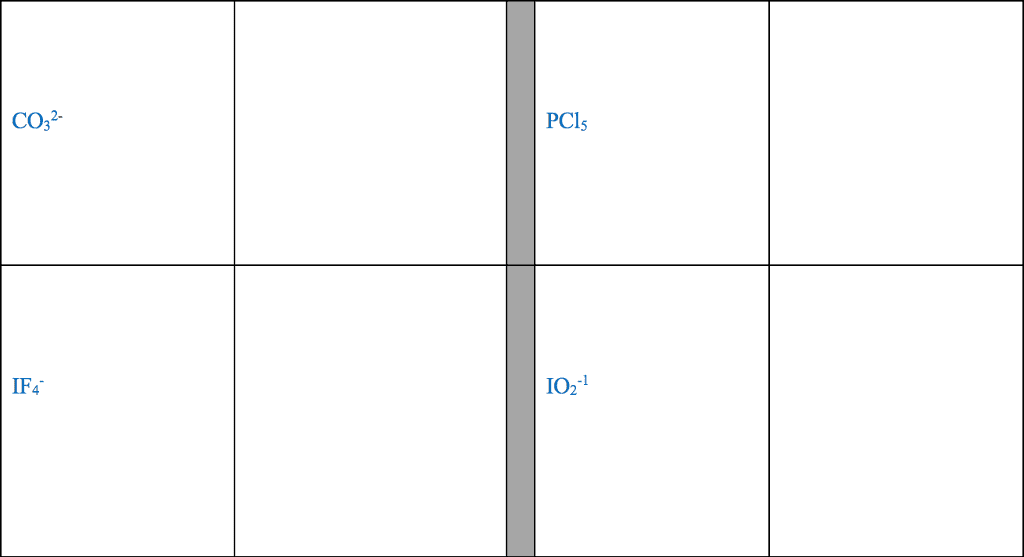
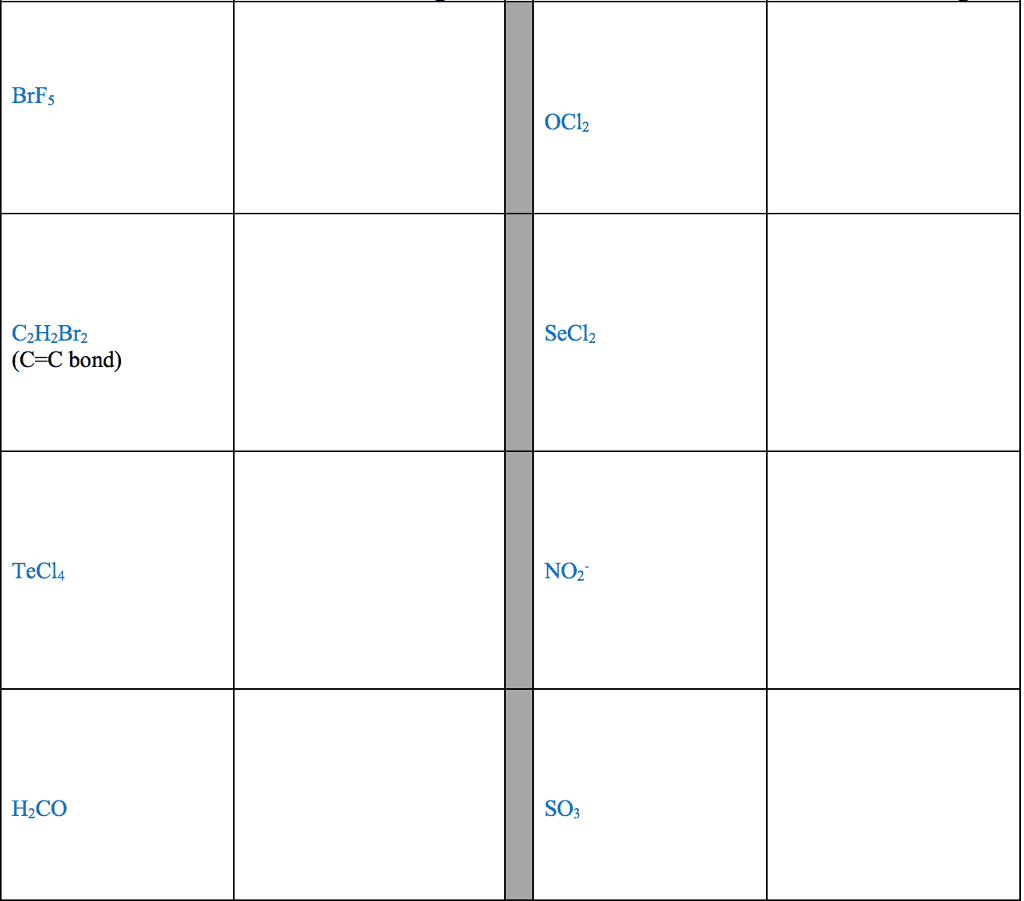
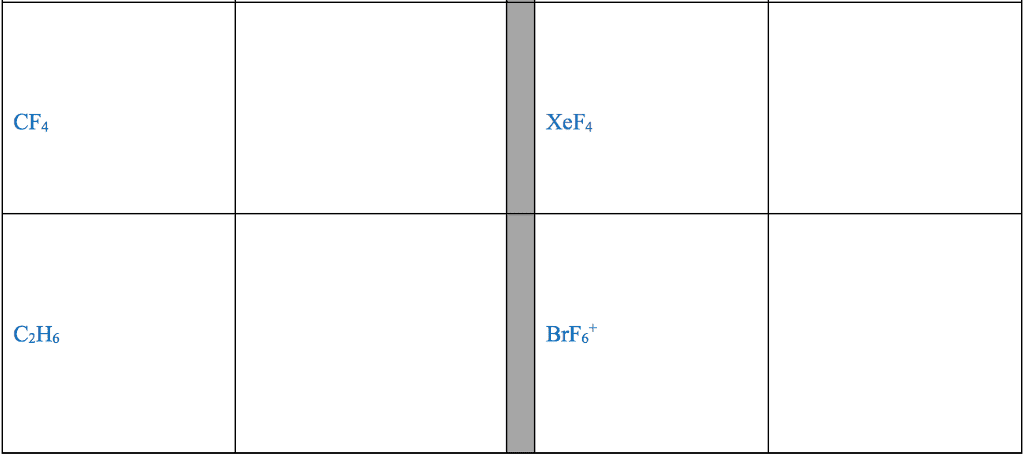
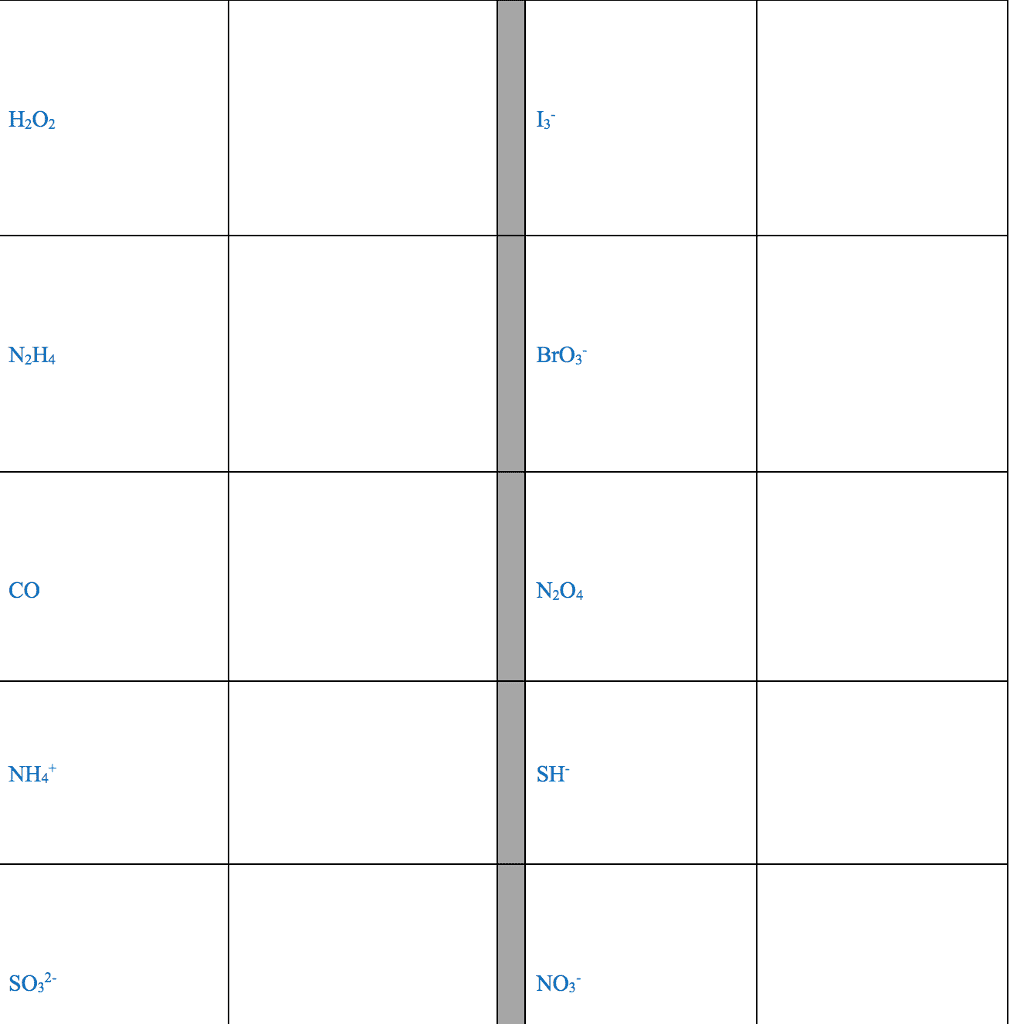
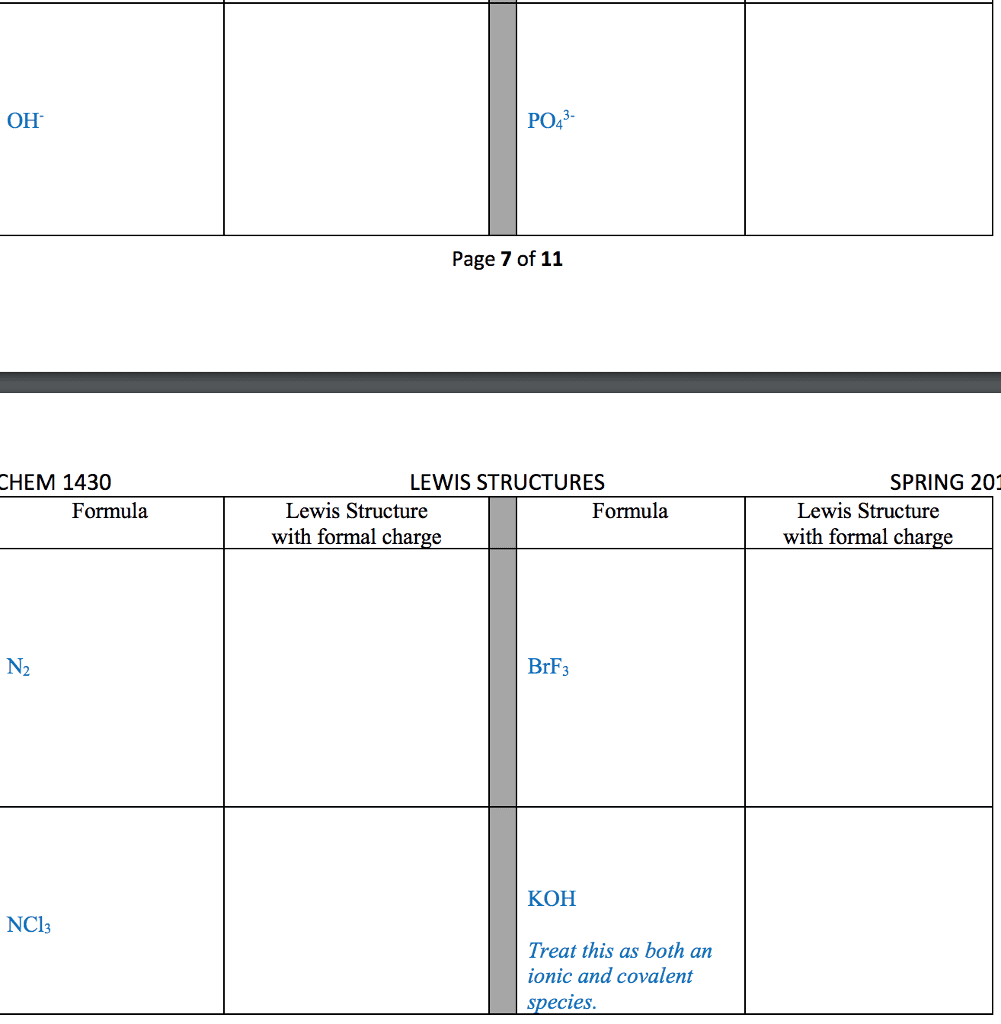
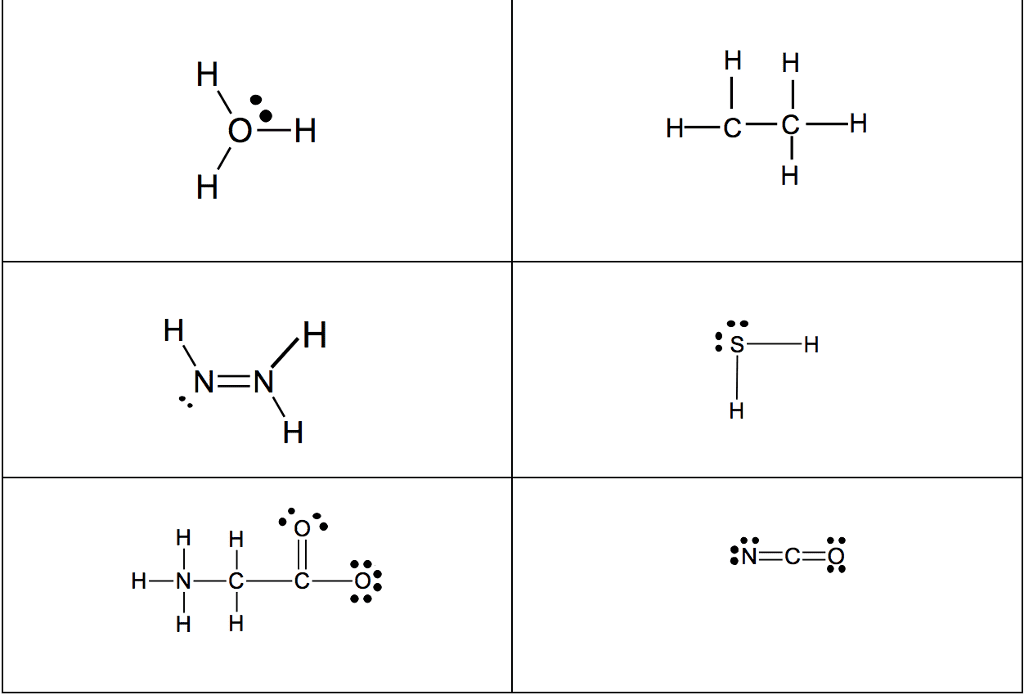
1. electrons, especially those of the outermost (valence) electronic shell, play a fundamental role in chemical bonding: in some cases, electrons are transferred from one atom to another. Positive and negative ions are formed and attract each other through electrostatic forces called ionic bonds: in other cases, one or more pairs of electrons are shared between atoms. A bond formed by the sharing of electrons between atoms is called a covalent bond: electrons are transferred or shared in such a way that each atom acquires an especially stable electron configuration. Usually this is a noble gas configuration, one with eight outer-shell electrons, or an octet. A lewis symbol consists of a chemical symbol to represent the nucleus and core (inner-shell) electrons of an atom, together with dots placed around the symbol to represent the valence (outer- shell) electrons. A lewis structure is a combination of lewis symbols that represents either the transfer or the sharing of electrons in a chemical bond.