CHEM 281 Chapter Notes - Chapter 10: Sn2 Reaction, Phosphoryl Chloride, Elimination Reaction
Document Summary
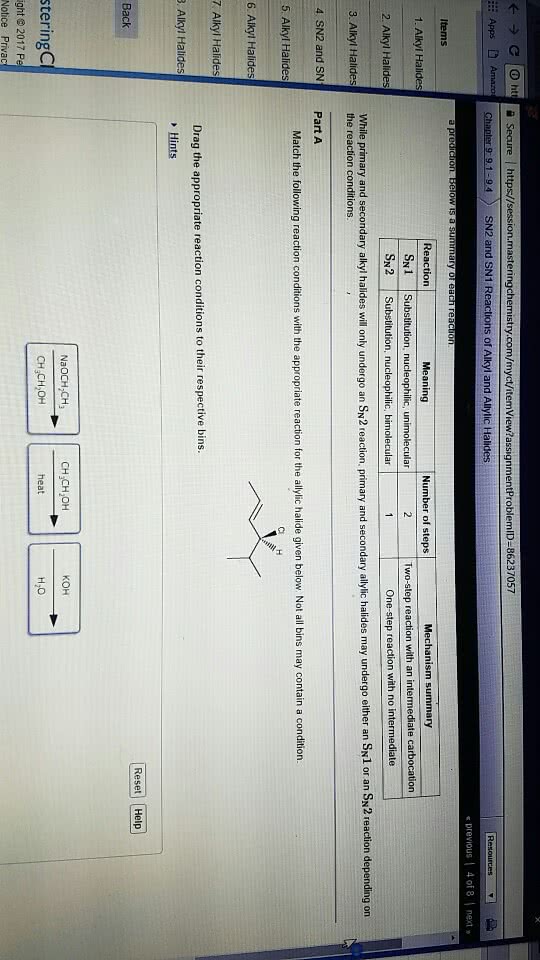
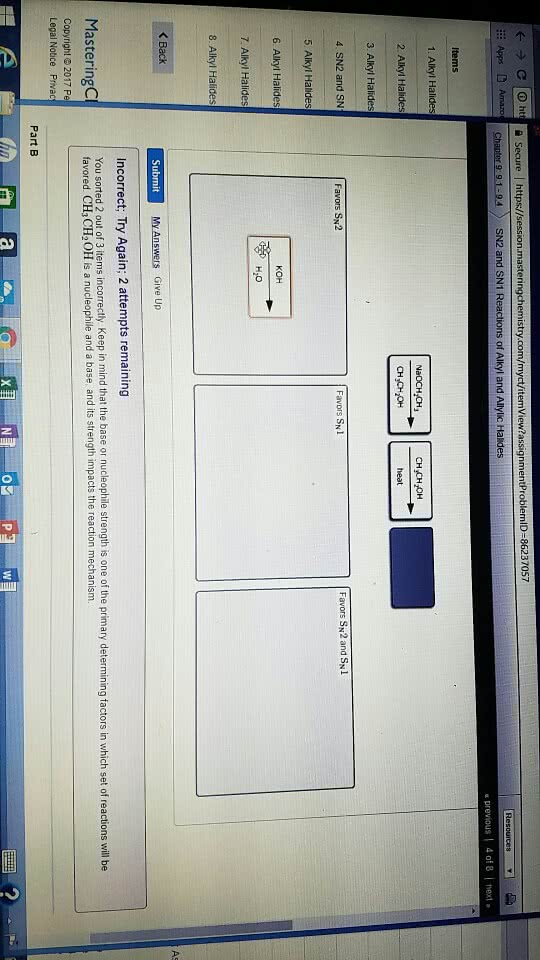
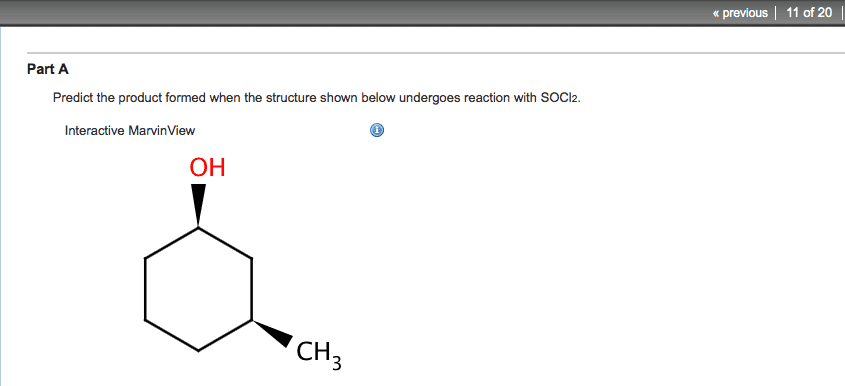
Alcohols undergo nucleophilic reactions with hi, hbr, and hcl to form alkyl substitution reactions with hi, hbr, and halides. Sn1 reactions in the case of secondary and. Sn2 reactions in the case of primary tertiary alcohols. alcohols. Epoxides undergo nucleophilic reactions with hbr or hi and heat; if departure substitution reactions. The leaving groups of alcohols and ethers are stronger bases than halide ions, so alcohols and ethers must be activated before they can undergo a substitution or elimination reaction. An alcohol can be activated by protonation. Therefore, it undergoes nucleophilic substitution reactions with hi, hbr, and hcl to form alkyl halides. These are sn1 reactions in the case of secondary and tertiary alcohols and sn2 reactions in the case of primary alcohols. Pcc, hypochlorous acid, and the swern oxidation oxidizes primary alcohols to aldehydes and secondary alcohols to ketones. Sn1 reactions form carbocation intermediates, so carbocation rearrangements can occur.