CHEM 1A03 Chapter Unit 7 Module 2: Chemistry Unit 7- Module 2 – Acid-Base Chemistry II

35
CHEM 1A03 Full Course Notes
Verified Note
35 documents
Document Summary
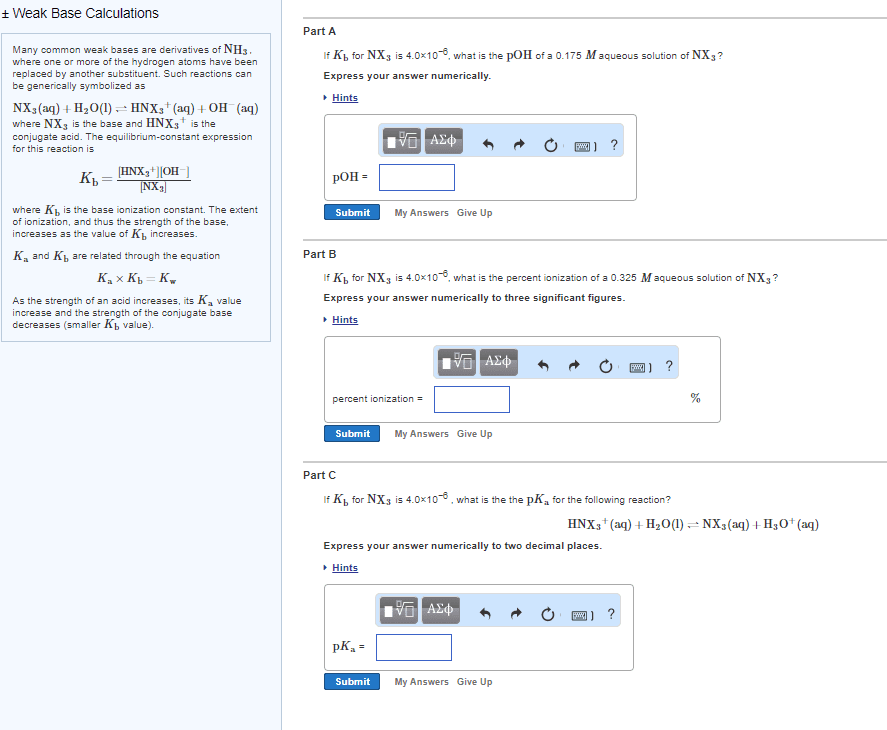
Chemistry unit 7: module 2 acid-base chemistry ii. A quantitative measure of the strength of an acid in solution. A quantitative measure of the strength of a base in solution. If we add the 2 reactions together, the total reaction would be 2 waters react to form h3o+ and oh- so the net equilibrium expression would simply be the concentration of h3o+ multiplied by the concentration of oh- When we add the two reactions together, we need to multiply the equilibrium constants together. If we take the negative log of both sides we get this expression: Log10(kw) = -log10(ka) + (-log10(kb)) pka & pkb relationship. A(cid:374)other (cid:449)ay of sayi(cid:374)g (cid:374)egati(cid:448)e log is usi(cid:374)g (cid:862)p(cid:863: pkw is pka plus pkb for a conjugate acid-base pair. Pkw is always equal to 14 at 25 degrees celcius. If we have ka and need kb, you can also use the relationship kw = ka x kb (avoids converting to pka and pkb)