CHEM 212 Chapter Notes - Chapter 1: Acetylene, Valence Electron, Lewis Structure
Document Summary
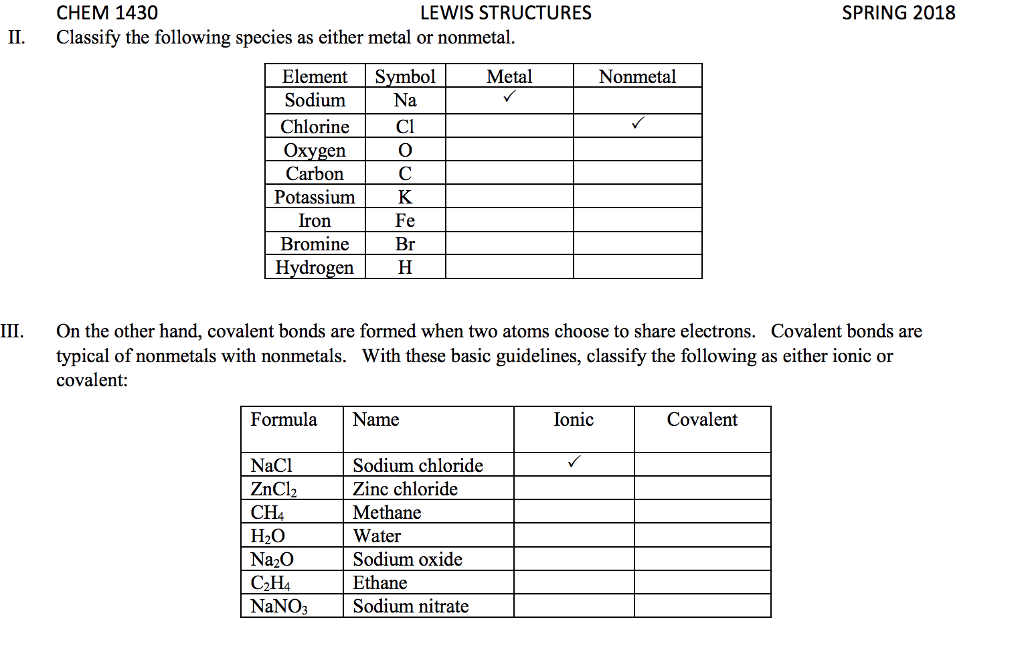
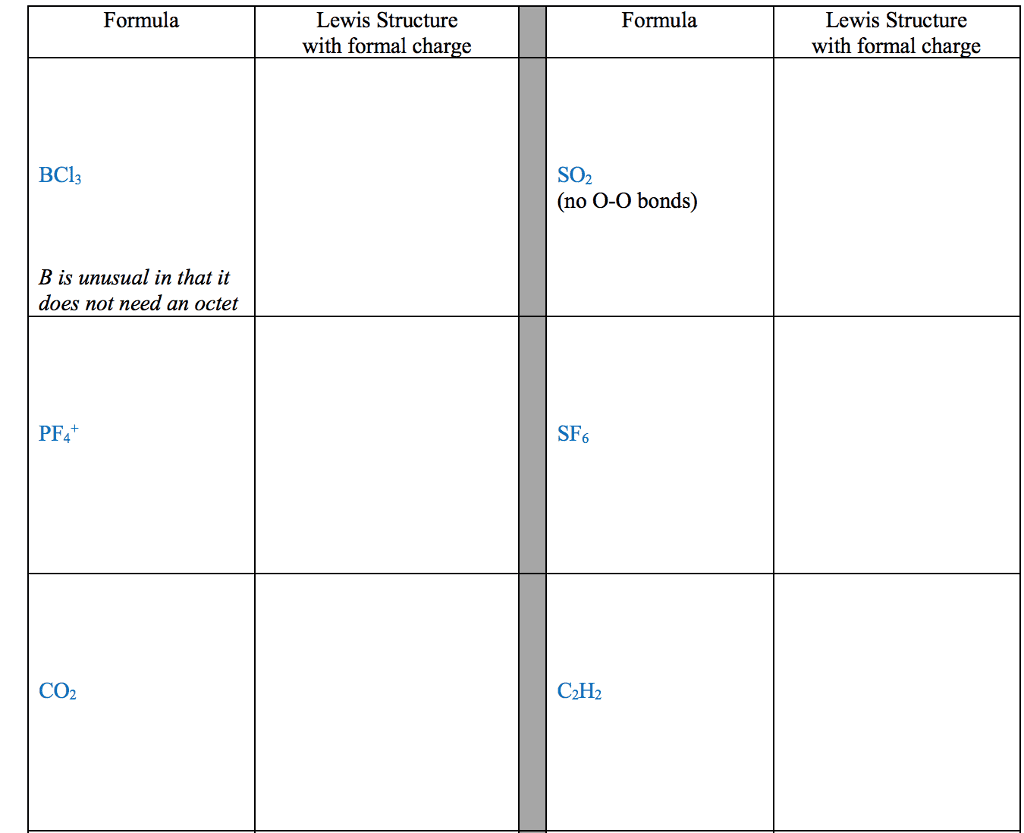
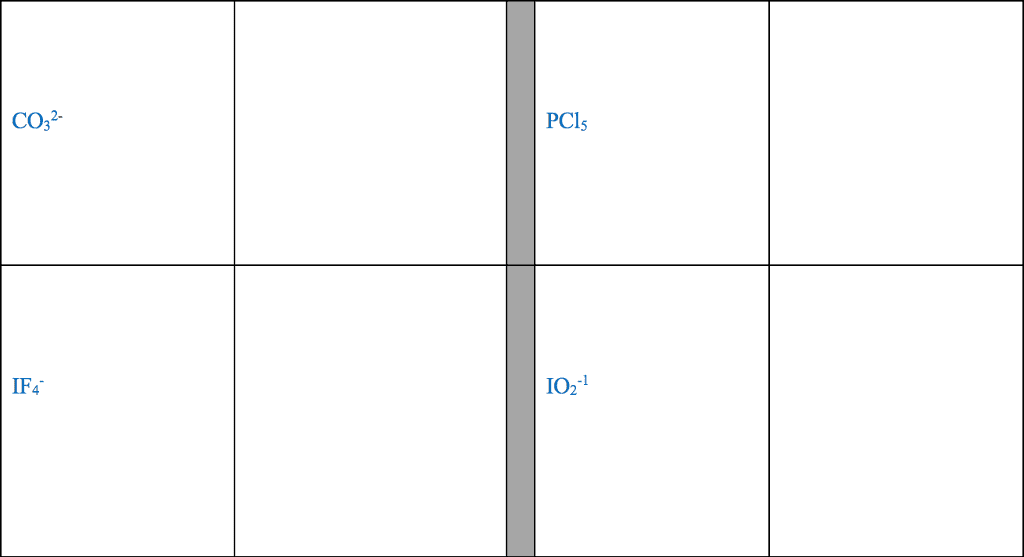
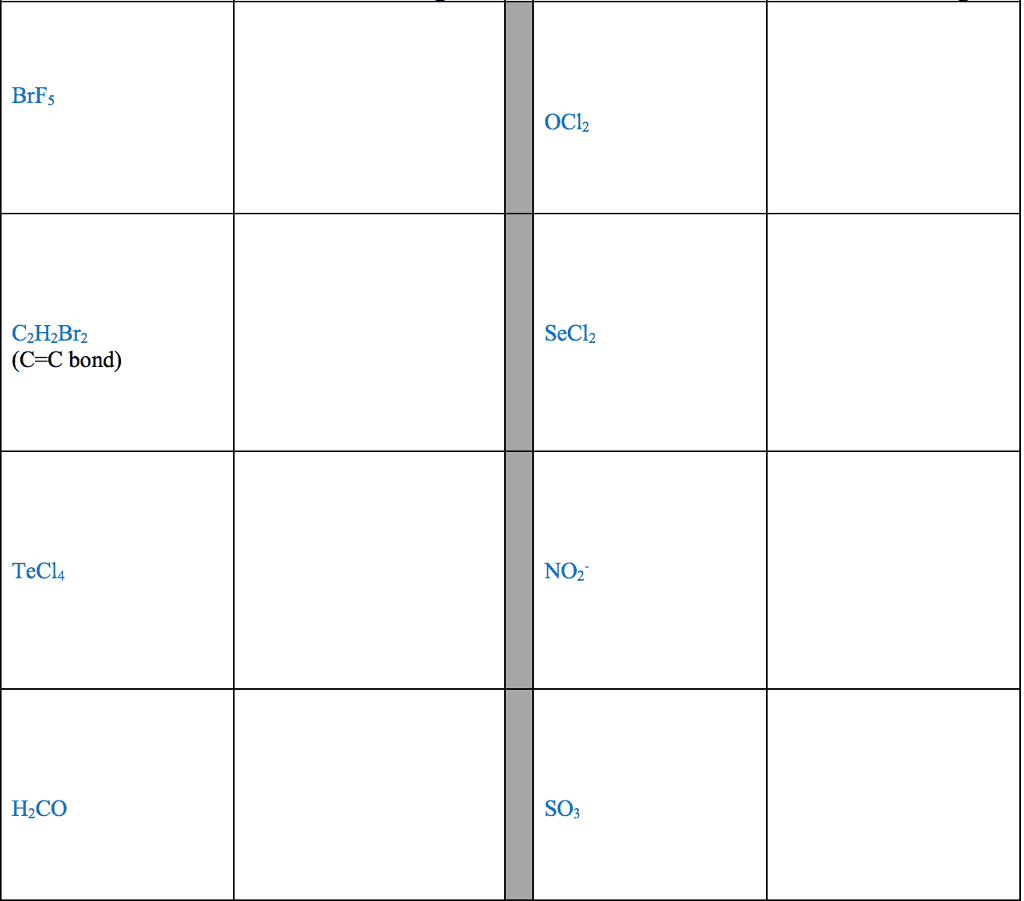
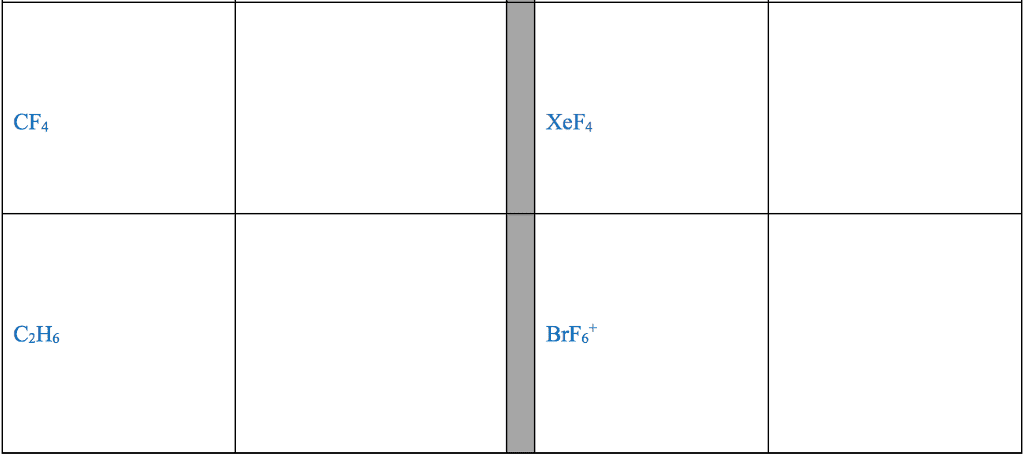
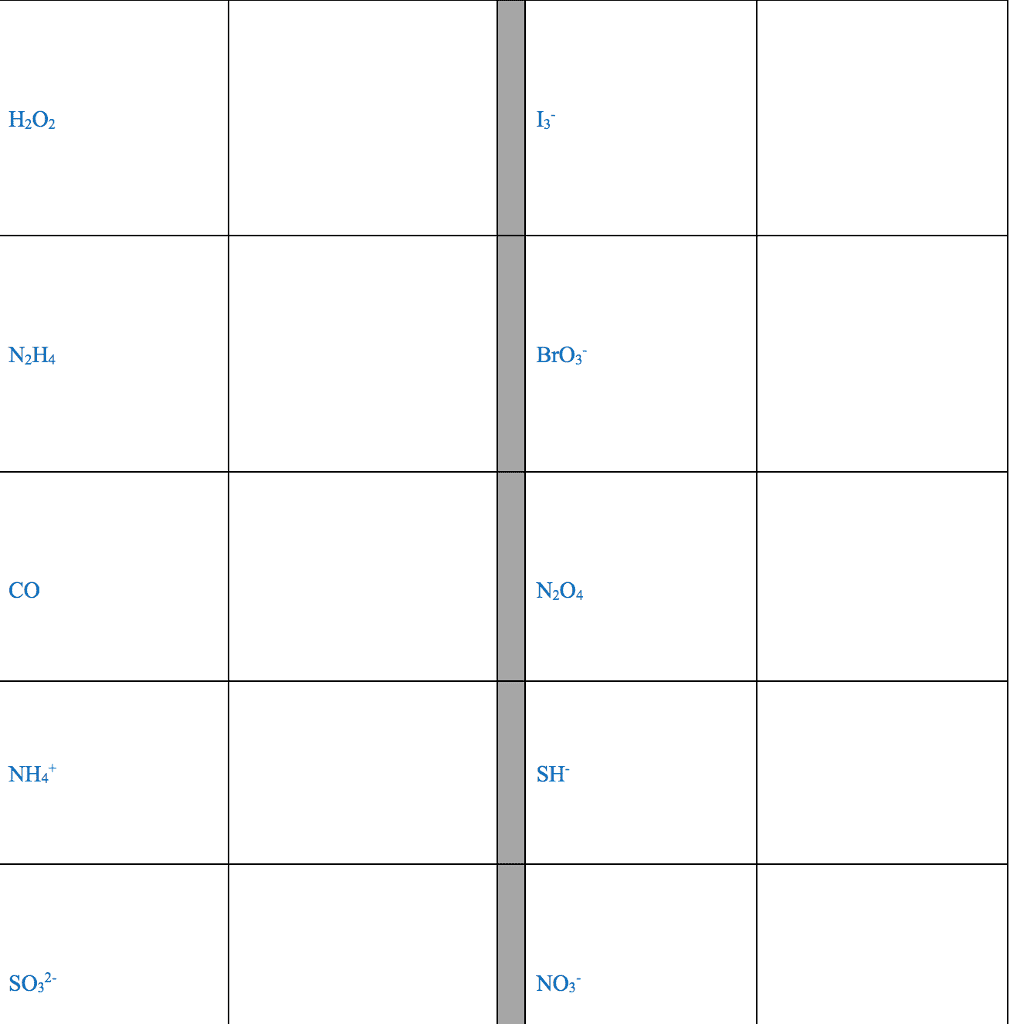
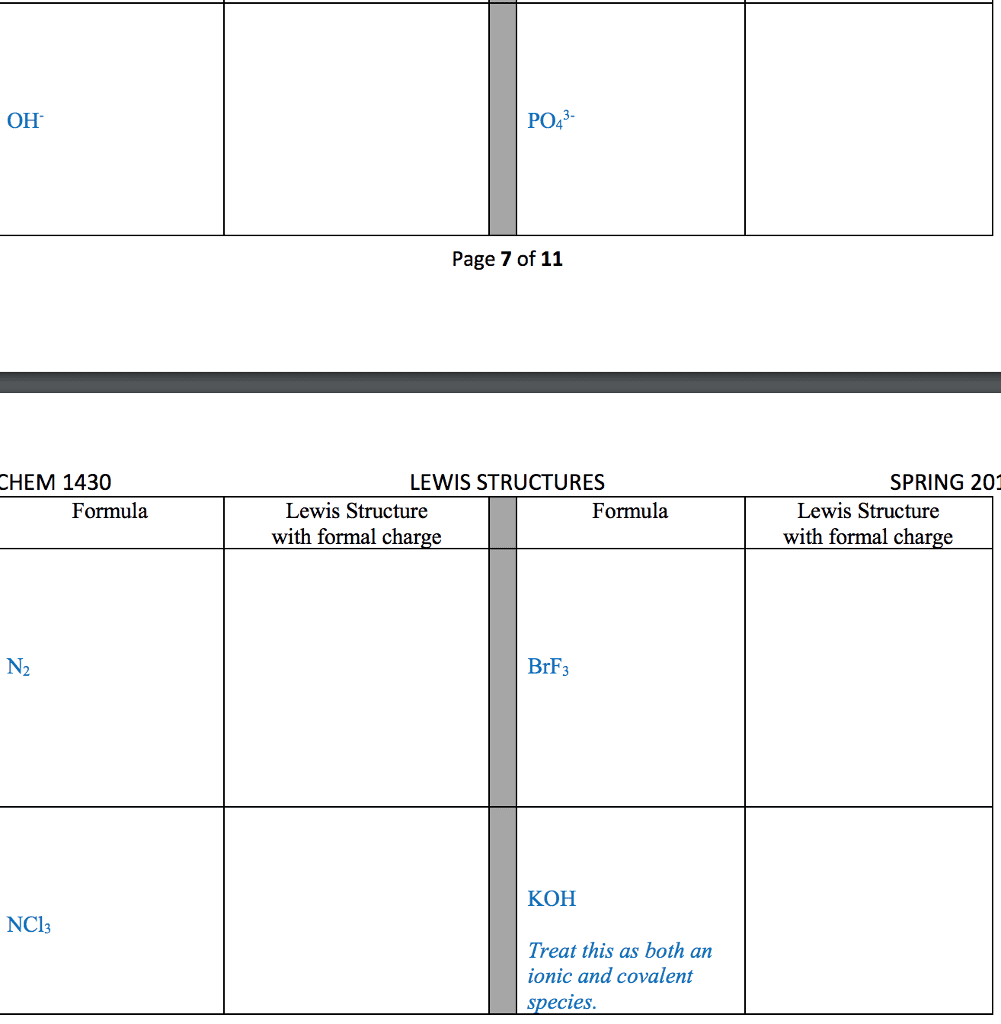
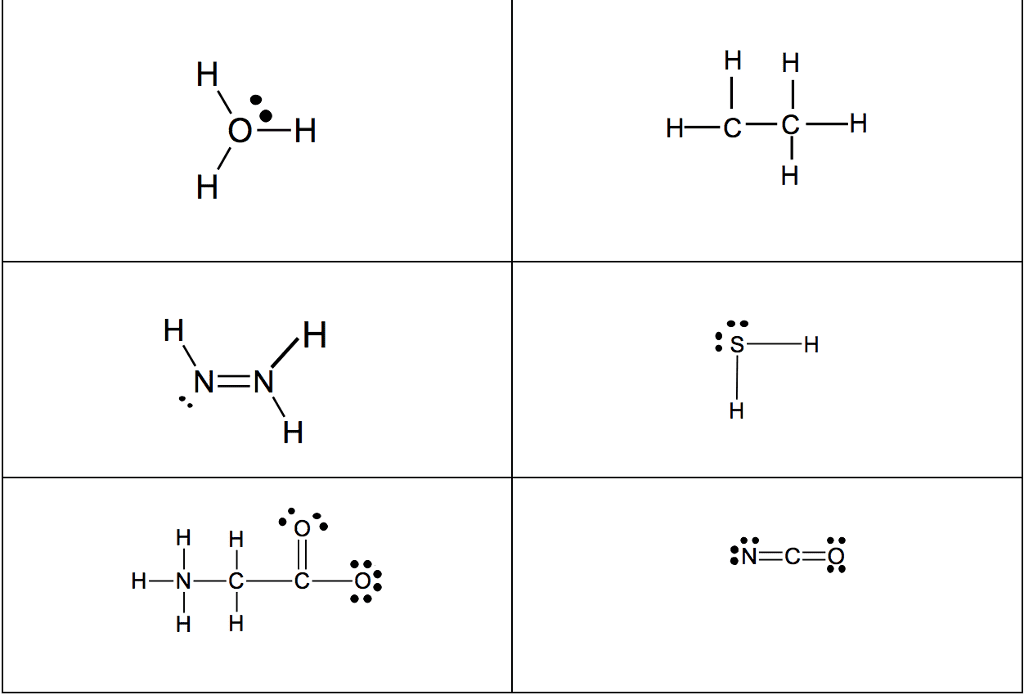
Several simple rules allow us to draw proper lewis structures: lewis structures show the connections between atoms in a molecule or ion using only the valence electrons of the atoms involved. If the structure is a positive ion (a cation), we subtract one electron for each positive charge: in drawing lewis structures we try to give each atom the electron configuration of a noble gas. The following problems illustrate the rules above: if necessary, we use multiple bonds to satisfy the octet rule (i. e. , give atoms the noble gas configuration). The organic molecules ethene (c2h4) and ethyne (c2h2), as mentioned earlier, have a double and triple bond, respectively: before we can write some lewis structures, we must know how the atoms are connected to each other. Even though the formula for nitric acid is often written hno3, the hydrogen is actually connected to an oxygen, not to the nitrogen.