CHEM 0N01 Chapter Notes - Chapter 3: Atomic Orbital, Atomic Number, Atomic Radius
Document Summary
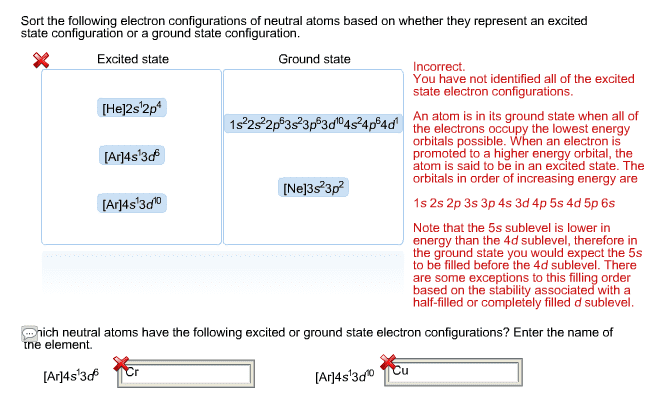
The electron that distinguishes an element from the previous element (with respect to atomic number) must enter the lowest energy atomic orbital available: fill from lowest to highest energy. Electrons must be distributed among orbitals of equal energy such that as many e- remain unpaired. *in order of n (energy levels):[ar] 3d6 4s2: br 35e, complete, condensed: *in order of n (energy levels):[ar] 3d10 4s2 4p5. Electrons has both wave + particle properties (wave/particle duality) Location of electrons described by schrodinger"s equation (solution to this equation define a volume in space (orbital) there is a 90% probability of locating an electron) The address of an electron can be described using quantum numbers. Quantum numbers: principle quantum number (n): n=1 to . Indicates the energy level (shell) of an atomic orbital. *max # of e- is an energy level = 2n2: n = 1, n = 2, n = 3, n = 4.