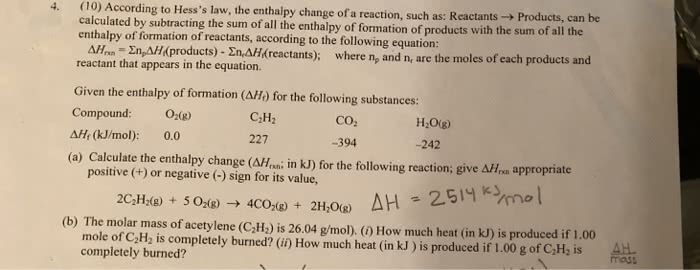University College - Chemistry Chem 112A Study Guide - Quiz Guide: Enthalpy, Heat Capacity
Document Summary
Kirchoff"s law: h t2 = h t1 + cp(t2-t1: used to calculate an enthalpy at a specific heat using the . H of a reaction at a different state. H (molar enthalpy) = n(bonds breaking) n(bonds formed) Enthalpy of formation: formation of 1 mole of molecule from elements in standard states: standard states: most stable form of the pure element at 1 atm and 298 k. Molar enthalpy: enthalpy associated with breaking 1 mol of bonds in gas phase. Enthalpy of atomization: enthalpy change associated with creating. 1 mol gas phase from element in standard state. Enthalpy of reaction: enthalpy change for chemical reaction in which all reactions & products are in standard state. Hess"s laws: add h (enthalpy) of reactions for overall new reaction (since it"s a state function path doesn"t matter)