CHEM 1035 Final: Final Exam - Fall 2017
Document Summary
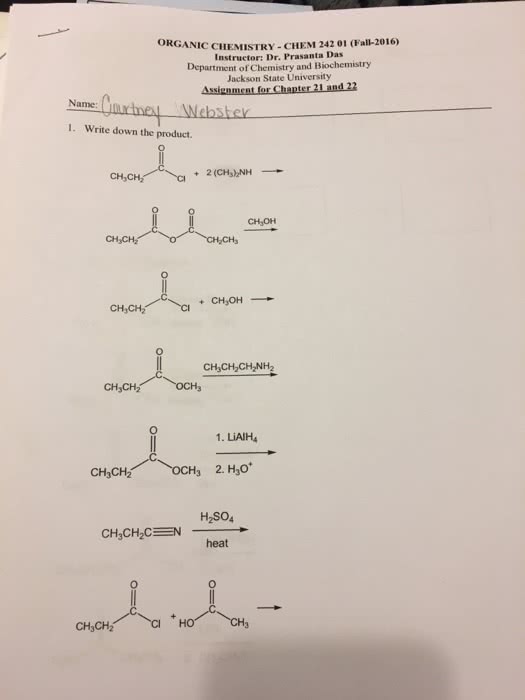
Select the correct lewis structure for nitrogen trifluoride, nf3: answer: a, bond order = shared electron pairs / bonded-atom pairs. For the resonance structures above, there are 4 shared electron pairs and 3 bonded- atom pairs. Formal charges: the most important structures are ones where the formal charges are closest to zero, if they are not all zero, the more electronegative atom is preferred to have a negative formal charge. In the diagram above, the structures that are circled are always nonpolar if the terminal atoms are identical. In the structure above, the bonds are polar, but the individual bond polarities cancel. In the structure above, the bonds are polar, and the bond polarities do not cancel. The shared space is occupied by 2 electrons, which have opposite spins: overlapping region: electron spends most of their time between the two nuclei, orbital orientation and maximum overlap. The greater the extent of orbital overlap, the stronger the bond: hydrogen fluoride: