CHEM 104 Study Guide - Midterm Guide: Conjugate Acid, Oxoacid, Chief Operating Officer
Document Summary
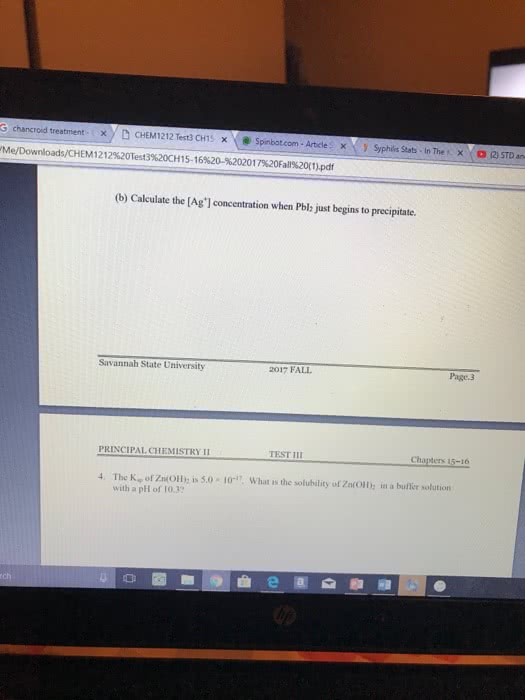
If slow step comes first then rate law just comes straight from first step. If rate law is not the first step, rearranging will have to be done. An intermediate is a substance produced and consumed within the steps of the . Slow step 1: 2 +2 3 +, rate = 1[2]2. Fast step 2: 3 +2 rate = 2[3] If the rate equation for the rate determining step is the same as the experimentally determined rate equation for the reaction then the rate is viable. There are finite concentrations of reactants and products, and these concentrations remain constant unless conditions change: an equilibrium always results in smaller amounts of products than the theoretical yield predicts. Reactant favored: when the equilibrium mixture consists mostly of reactants. When a chemical reaction reaches equilibrium and concentrations of reactants and products remain constant, it appears the has stopped. Dynamic equilibrium: the rate of the forward exactly equals the rate of the reverse .