Thanks for your help. Please show work and equations used.
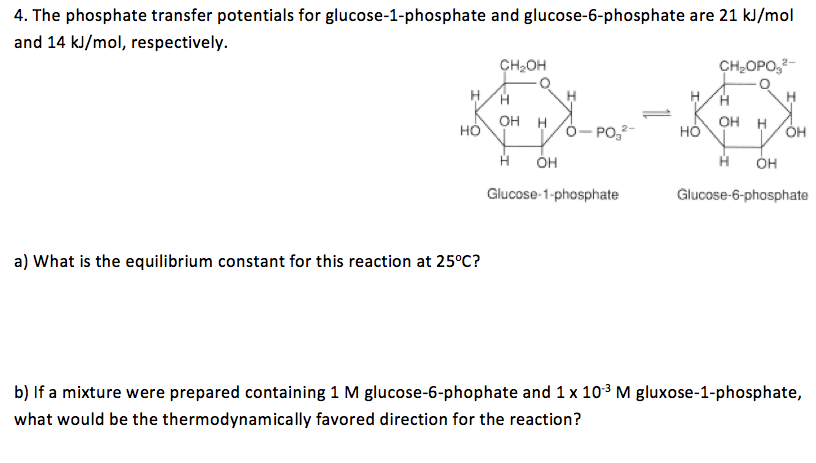
The phosphate transfer potentials for glucose-1-phosphate and glucose-6-phosphate are 21 kJ/mol and 14 kJ/mol, respectively. Glucose 1-phosphate Glucose-6-phosphate What is the equilibrium constant for this reaction at 25degreeC? If a mixture were prepared containing 1 M glucose-6-phophate and 1 x 10-3 M gluxose-1-phosphate, what would be the thermodynamically favored direction for the reaction?
Show transcribed image text The phosphate transfer potentials for glucose-1-phosphate and glucose-6-phosphate are 21 kJ/mol and 14 kJ/mol, respectively. Glucose 1-phosphate Glucose-6-phosphate What is the equilibrium constant for this reaction at 25degreeC? If a mixture were prepared containing 1 M glucose-6-phophate and 1 x 10-3 M gluxose-1-phosphate, what would be the thermodynamically favored direction for the reaction?
