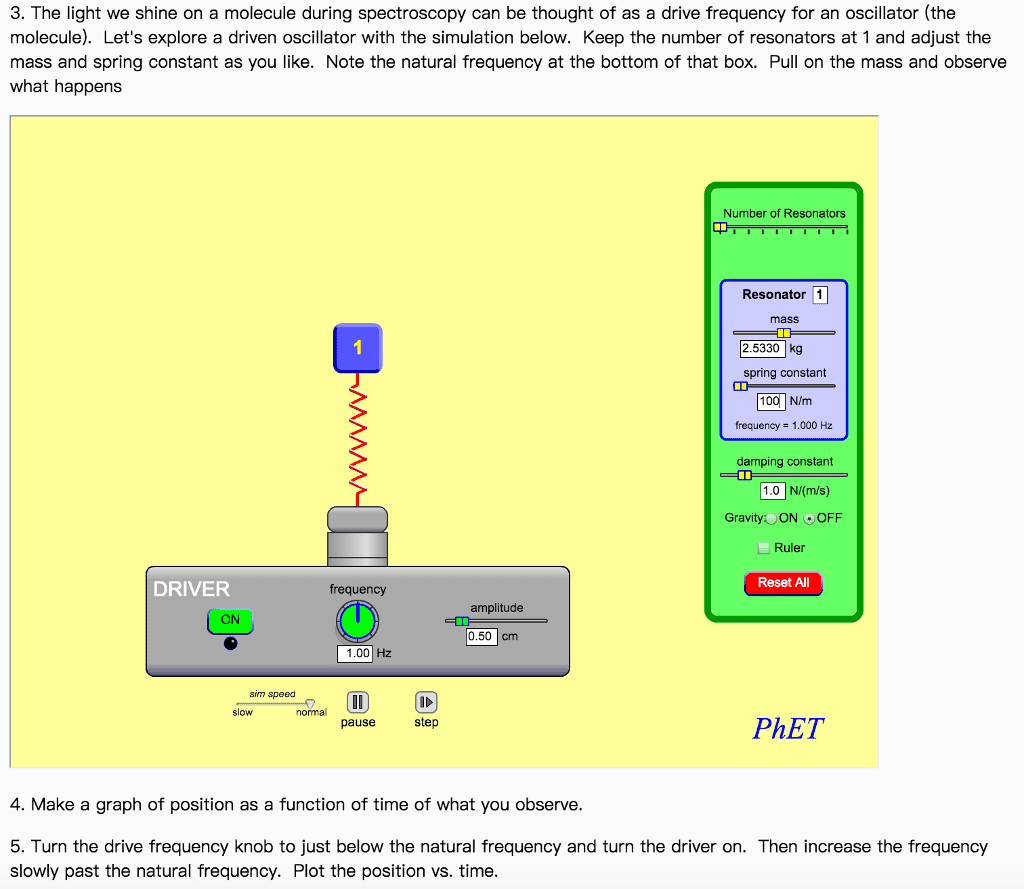
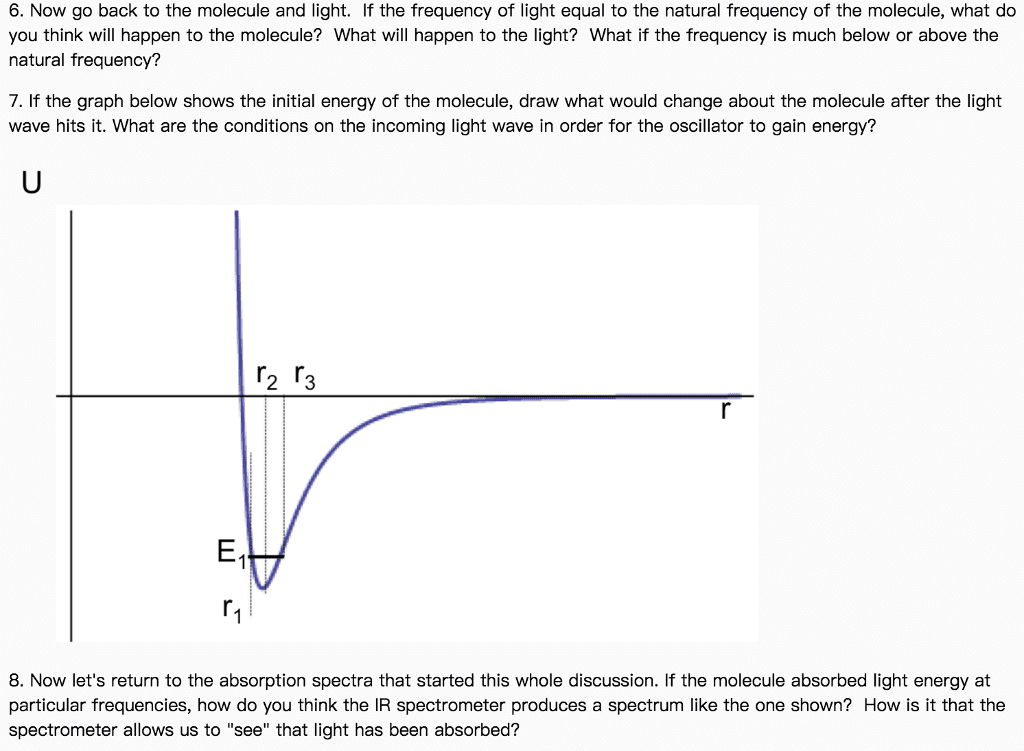
CHEM 152 Study Guide - Final Guide: Raman Spectroscopy, Raman Scattering, Rayleigh Scattering
Document Summary
Every molecule has unique vibrational and rotational motions -- molecular fingerprint. Ir and raman spectroscopy used to study this. Raman spectroscopy measures the scattered light; involves change in polarizability of the molecule during the molecular vibration; acquired via excitation by single wavelength (different raman shift observed corresponding to different vibrational levels in the molecule. Strongly ir active/ weakly raman active: vibrations of polar bonds; o-h, c+o, Weakly ir active/ strongly raman active: vibrations of less polar bonds or vibrations involving larger atoms (with weakly-held electrons); c=c, c-c, c-h, bonds involving cl. Anti-stokes weaker than stokes scattering -- main difficulty is to detect raman scattering by filtering out strong rayleigh scattering. Use multiple monochromators to selectively transmit needed wave range to reduce rayleigh intensity. Chem 152 page 1 range to reduce rayleigh intensity. Disadvantage: higher resolving power needed (10^5) while ir needs only 10^ Can be used in aqueous solutions (water can absorb ir light strongly and affect ir spectrum)