HNF 150 Study Guide - Midterm Guide: Nitrogen Balance, Amine, Blood Proteins
Get access

Related Documents
Related Questions
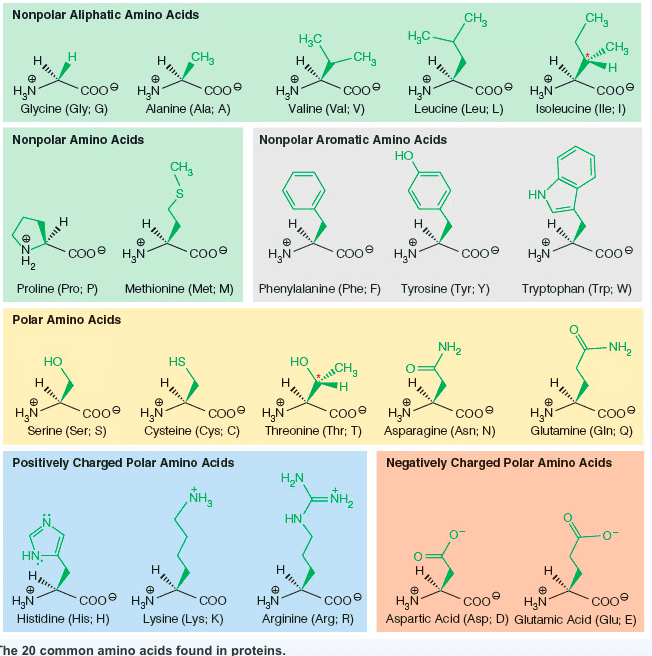
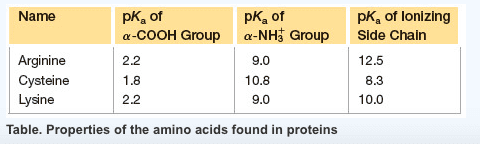
Using the information in the table below, enter the correct pH that should be used to separate the following three amino acids by electrophoresis:
(a) Glu, Lys, and Thr: pH=?
(b) His, Lys, and Arg: pH=?
------------------------------------------------------------------------------------------------------------------------------------------------------
| Amino Acid | pKa of α-COOH | pKa of α-NH3+ | pKa of Side Chain | Isoelectric Point (pI) |
|---|---|---|---|---|
| Alanine | 2.35 | 9.87 | - | 6.11 |
| Arginine | 2.01 | 9.04 | 12.48 | 10.76 |
| Asparagine | 2.02 | 8.80 | - | 5.41 |
| Aspartic acid | 2.10 | 9.82 | 3.86 | 2.98 |
| Cysteine | 2.05 | 10.25 | 8.00 | 5.02 |
| Glutamic acid | 2.10 | 9.47 | 4.07 | 3.08 |
| Glutamine | 2.17 | 9.13 | - | 5.65 |
| Glycine | 2.35 | 9.78 | - | 6.06 |
| Histidine | 1.77 | 9.18 | 6.10 | 7.64 |
| Isoleucine | 2.32 | 9.76 | - | 6.04 |
| Leucine | 2.33 | 9.74 | - | 6.04 |
| Lysine | 2.18 | 8.95 | 10.53 | 9.74 |
| Methionine | 2.28 | 9.21 | - | 5.74 |
| Phenylalanine | 2.58 | 9.24 | - | 5.91 |
| Proline | 2.00 | 10.60 | - | 6.30 |
| Serine | 2.21 | 9.15 | - | 5.68 |
| Threonine | 2.09 | 9.10 | - | 5.60 |
| Tryptophan | 2.38 | 9.39 | - | 5.88 |
| Tyrosine | 2.20 | 9.11 | 10.07 | 5.63 |
| Valine | 2.29 | 9.72 | - | 6.00 |
| Amino Acid | M.W. g/mol | pKa for side chain |
| Alanine | 89.1 | |
| Arginine | 174.2 | 12.48 |
| Asparagine | 132.1 | |
| Aspartate | 133.1 | 3.90 |
| Cysteine | 121.1 | 8.37 |
| Glycine | 75.0 | |
| Glutamate | 147.1 | 4.07 |
| Glutamine | 146.1 | |
| Histidine | 155.1 | 6.04 |
| Isoleucine | 131.2 | |
| Leucine | 131.2 | |
| Lysine | 146.2 | 10.54 |
| Methionine | 149.2 | |
| Proline | 115.1 | |
| Phenylalanine | 165.2 | |
| Serine | 105.1 | |
| Threonine | 119.1 | |
| Tryptophan | 204.2 | |
| Tyrosine | 181.2 | 10.46 |
| Valine | 117.1 |
Take a look at your pKa table for the amino acids. It turns out that when these side chains are in a protein, they can have very different pKa values. Explain the following observations:
a. In the enzyme medium chain acyl-CoA dehydrogenase (b-oxidation of fatty acids), E376 has a measured pKa ~ 8.0. It is found in a very hydrophobic environment. What could account for it elevated pKa?
b. For the enzyme aconitase (Krebbâs Cycle), a catalytic serine is known to have a pKa ~ 8. What kind of factors could account for this decrease in pKa?