CHEM 1332 Study Guide - Final Guide: Labatt Brewing Company, Molar Mass, Boiling Point
34 views21 pages
11 Aug 2020
School
Department
Course
Professor
Document Summary
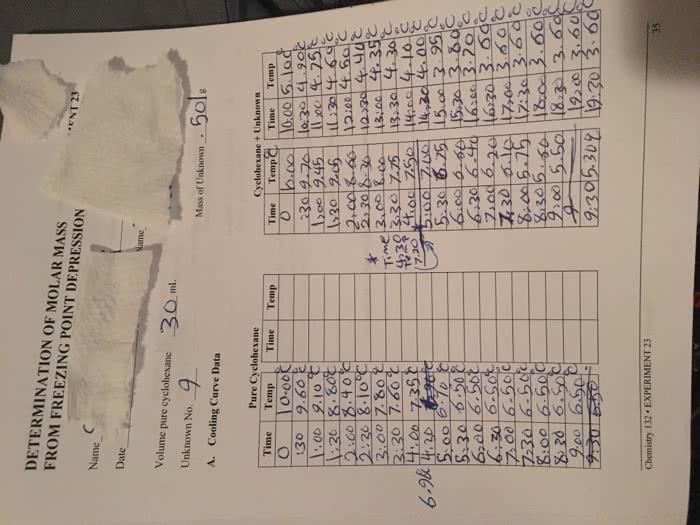
Labatt a. to#rgpenies:scolllgative only on the solute concentration property : physical property that depend at = tsoiv - tsolution dtf = i ktm. *knowthis : mm = #kf du freezing : subtract ( tdf ) ( mass solvent : kg ) boiling : add. It thermometer reads a different temp : no effect on molar mass. Freezing point of electrolyte ( t vhf) so if will i be more significant. * a solution has a lower freezing temperature than a pure solvent ; the more concentrated a solution becomes the lower thefreezing point. O *dated (cid:12200) crystallizes pamgoieutgrie rated as it freezes cooling down over time ask. mu#ifaetteenwsucgnnttoma/&eyosmiifm:gie ine3 n. Lower freezing pt than pure subst . t. at#i4ikinetics-n-uolkrxh:. ralereauton:- concentration of a product or reactant with respectto time. Iz t starch blue 1 black complex ionic strength s % % ennmafa *dan rate constant in .
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232