CHEM 1113 Study Guide - Final Guide: Trailing Zero, Decimal Mark, Subatomic Particle
191 views7 pages
Document Summary
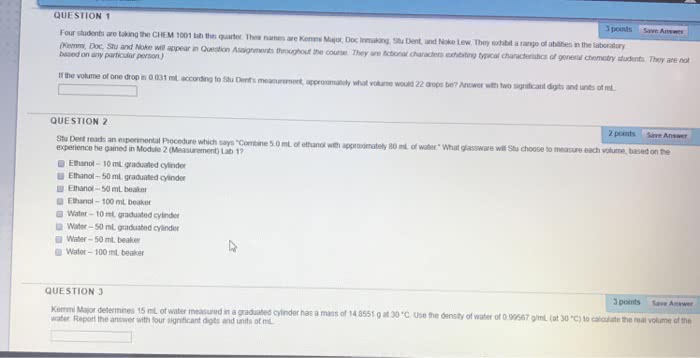
Chem 1113 fall 2016 exam 1 study sheet. Chapters 1 and 2 - math and concept review concept area 1 - metric system: using the information about metric prefixes, symbols and multiples, you should be able to convert between units. Know these prefixes: you should be able to identify the common metric units for length, mass, temperature, volume and density. Si units: length = m, mass = kg, temp = kelvin (k, volume = l or ml (1 ml = 1 cm3, density= kg/m3 but usually g/ml = g/cm3. Concept area 3 - significant figures: be able to report measurements to the correct number of decimal places. The following abilities need to be demonstrated in handling significant figures. Determining which digits are significant: non-zero digits are always significant, leading zeros are not significant, confined zeros are always significant, trailing zeros are zeros at the end of a number. Always significant if there is a decimal point.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232