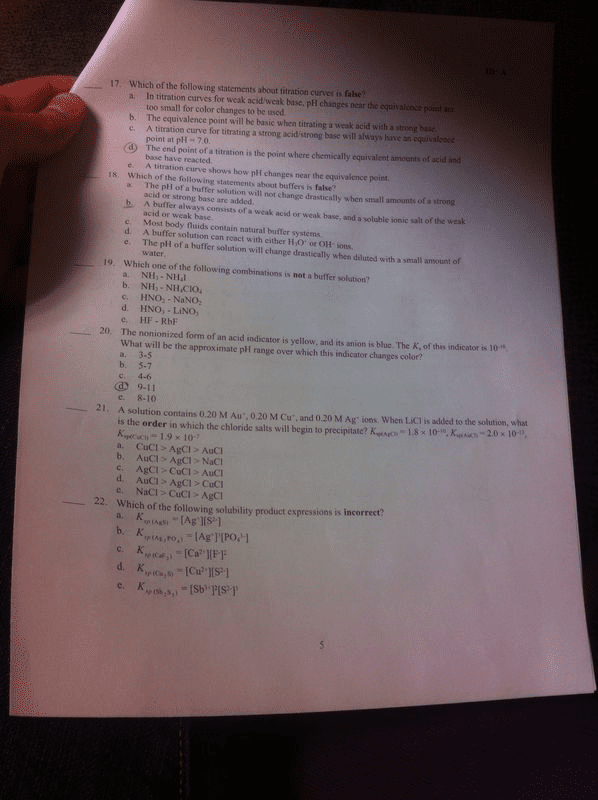CHEM 1A Study Guide - Midterm Guide: Titration, Conjugate Acid, Acid Dissociation Constant
28 views1 pages
Document Summary
Acid - base indicator: an indicator changes color with ph because it is form ( h in ) , and another color in its conjugate base form ( in - ) a weak acid that has one color in its acid. State have to be in different color. H ih cag ) t h2o c e , t h , ot lag , Cag , ka = end point : when the concentrations of its acid and base forms are equal. Poly protic acid titrations: many stoichiometric points. Initially naoh reacts completely with the acid to form the di proto conjugate base h . Cag , hz at cag) t hz o ce: at buffer region 1. Ph determined by the proton transfer equilibrium between hsa" ions and hs a " ions produced in the titration. System is at s , while all hs at lost their h.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232