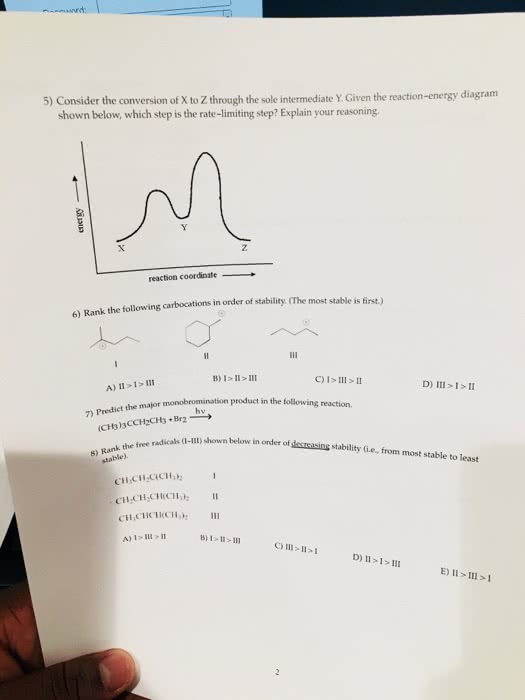
CHM 101 Study Guide - Final Guide: Free-Radical Halogenation, Unpaired Electron, Ultraviolet
Document Summary
Free radical halogenation is a reaction that substitutes a chlorine or a bromine for a hydrogen on an alkane. That is, it occurs only when performed in the presence of uv light (abbreviated hv). Typically, free radical reactions are described in three steps: initiation steps, propagation steps, and termination steps (described below). Note the use of a single headed arrow when describing the movement of a single electron. The reaction begins with an initiation step, which is the separation of the halogen (x2) into two radicals (atoms with a single unpaired electron) by the addition of uv light. This is called the initiation step because it initiates the reaction. The initiation step, or the formation of the chlorine radicals, is immediately followed by the propogation steps--steps directly involved in the formation of the product. As an example, isobutane (c4h10) will be used in the chlorination reaction. This first propogation step forms the tertiary radical.