CHM 2120 Study Guide - Quiz Guide: Trigonal Planar Molecular Geometry, Ethylamine, Linear Combination Of Atomic Orbitals
Document Summary
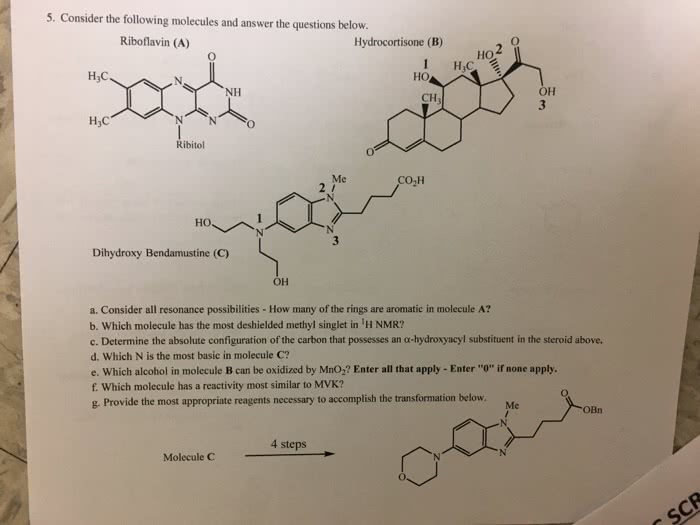
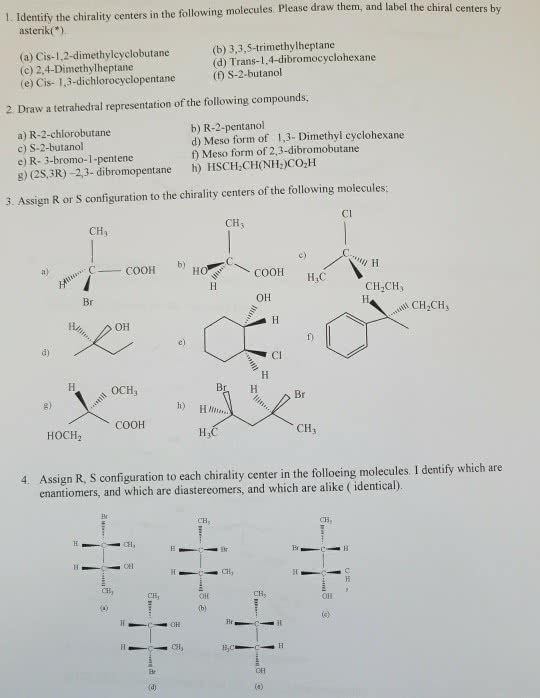
Assignment #1c - answer key: draw lewis structures, showing all unshared electrons, for the following molecules: (a) ch3nh2. N: there is a small portion of the periodic table that you must know to do organic chemistry. Construct this from memory including the group numbers, numbers of valence electrons, and electronegativities. Note: depending on the source you use, small differences exist between electronegativity values. Nevertheless, the relative values for electronegativity are always the same: draw structures for (a) two compounds with the formula c4h10. 2,2-propanediol (b) three compounds with the formula c3h8o2. 1,1-propanediol (check with your ta if you have drawn other structures and aren"t sure if they are correct) (c) two compounds with the formula c2h7n. C: c-b, name all the compounds in question 3. Answers in red: show the direction of the dipole moments of the following bonds. Br: n-h, o-h h) c-br, draw the shape of s and p orbitals including phasing.