CHEM 1300 Study Guide - Midterm Guide: Exothermic Reaction, Reaction Rate, Collision Theory
Document Summary
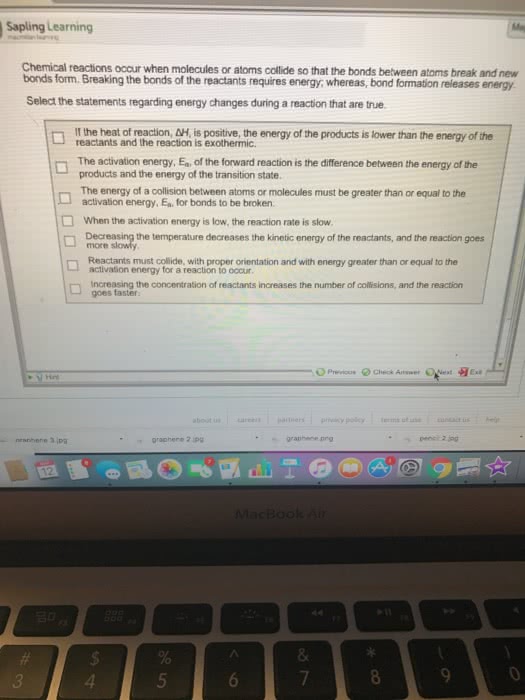
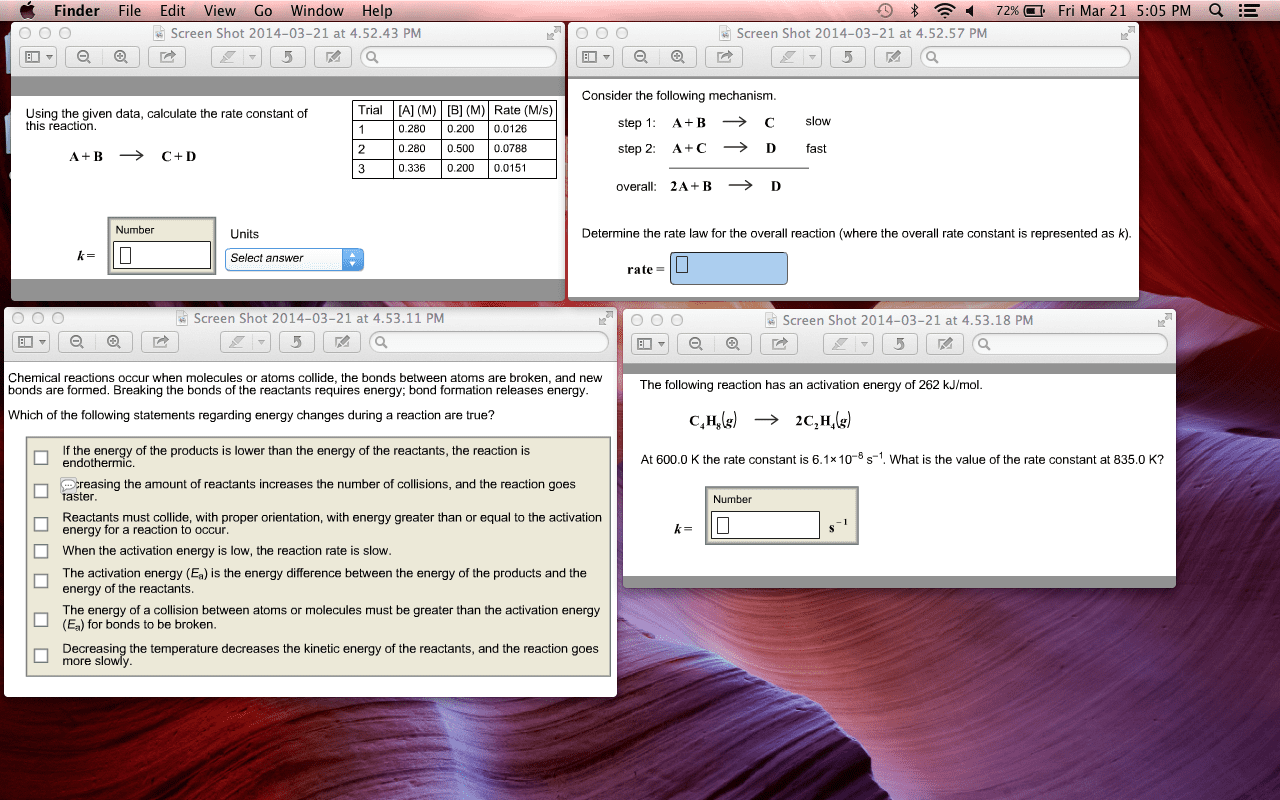
Kinetics is the branch of chemistry that studies the speed or rate which chemical reactions occur. Reaction rate: chemical reaction is one in which a new substance is formed. Growth of a plant: fast reactions will have short reaction time, and slow reactions will have long reaction time. Particles have to react under 2 requirements: right angle (orientation or geometry) - particles must collide at the proper orientation or geometry in order to collide effectively. Enough (activation energy) - particles must collide with enough energy to cause reaction. It refers to the speed with which a chemical reaction takes place. Temperature change over time: as the reaction proceeds the system will increase in temperature, the faster heat is produced, the higher the rate. As the reaction proceeds, for gaseous c is produced. This increases the pressure of the system, the faster the pressure increases, the greater the rate. As the reaction proceeds, reactants are used up and converted to gas.