CHEM 1A03 Study Guide - Midterm Guide: Enthalpy, Thermodynamics, Activation Energy

35
CHEM 1A03 Full Course Notes
Verified Note
35 documents
Document Summary
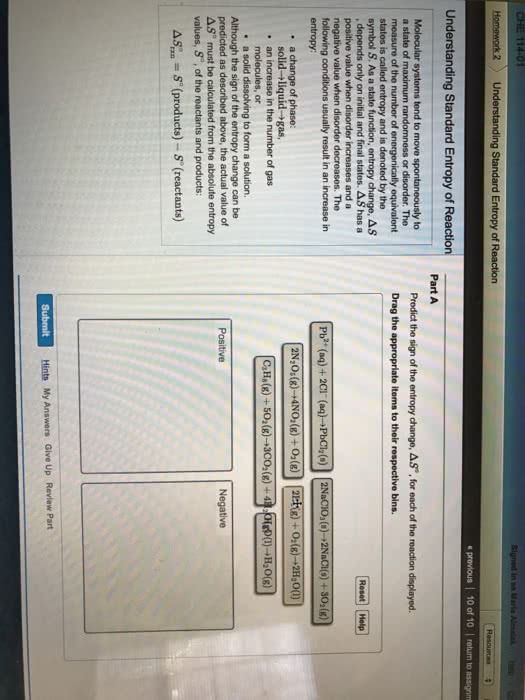
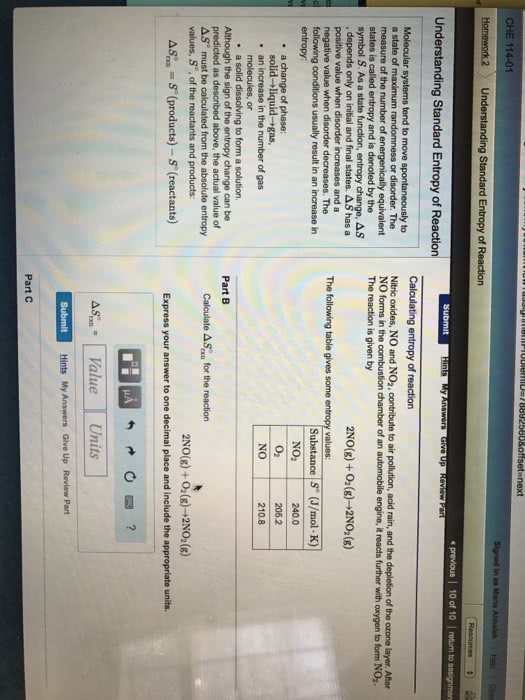
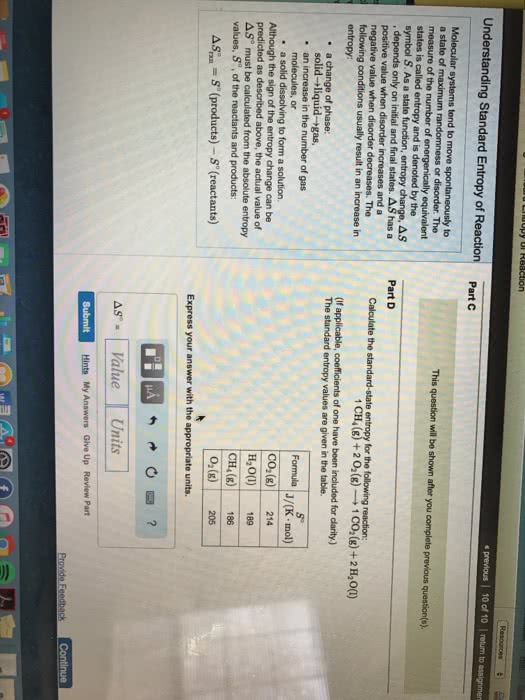
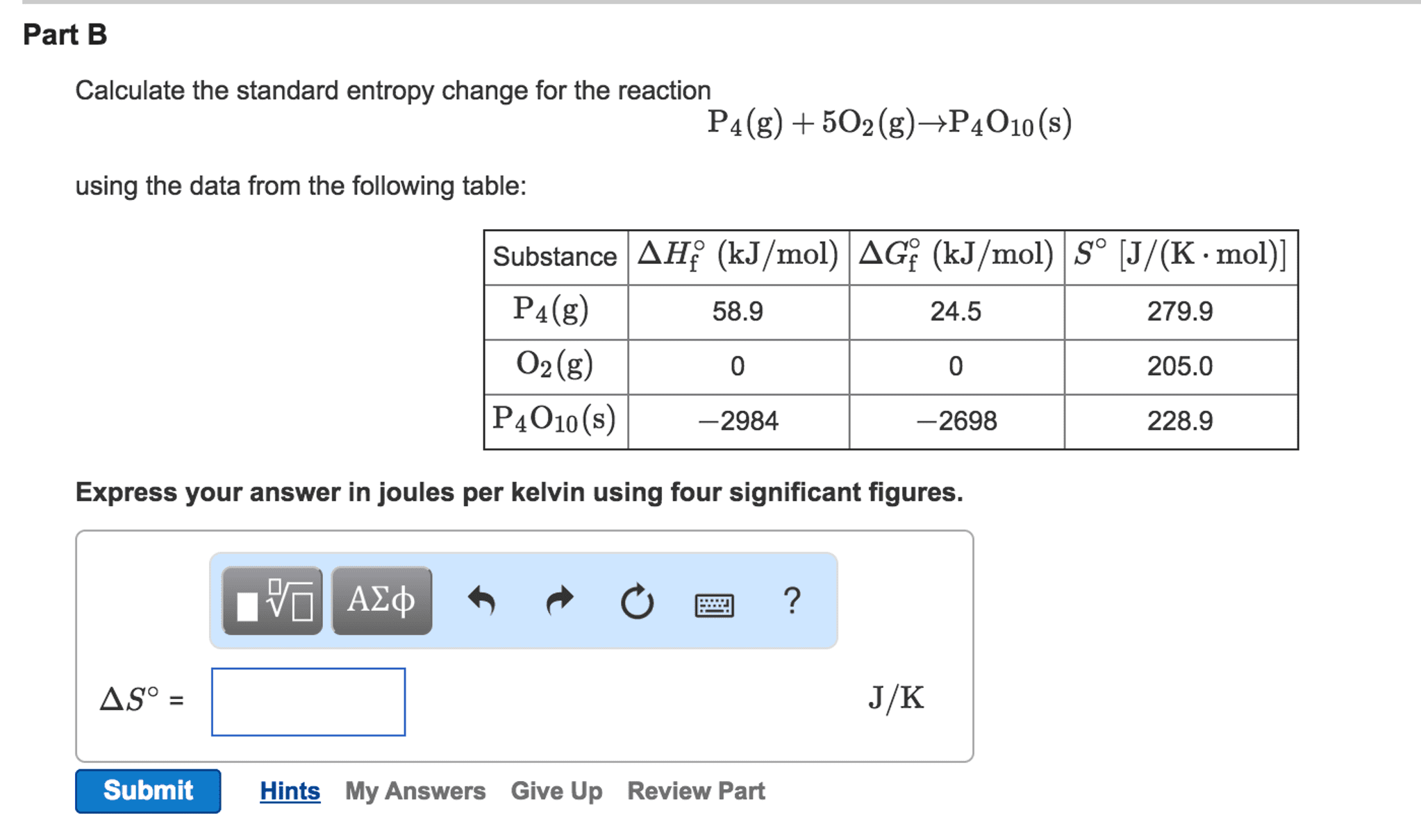
Entropy is a measure of the randomness or disorder of a system, or its surroundings, where chemical systems tend toward more disorder. Occurs when system becomes less disordered (more ordered) In general, a system will experience an increase in entropy ( s > 0) if. A mixture forms from pure substances the volume of the system increases the temperature increases the physical state of a substance changes. In chemical reactions, entropy increases ( s > 0) when. Complex molecules are broken down to simpler subunits. The value of entropy change, so, can be determined similar to the enthalpy change: reactants are at lower entropy state than products. This calculation uses entropy values, so, for substances, not entropy of formation values. All changes either directly or indirectly increase the entropy of the universe. Some reactions proceed without continuous outside assistance, given the necessary activation energy.