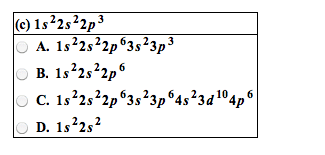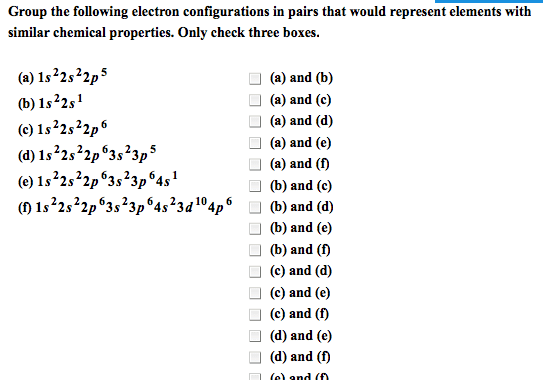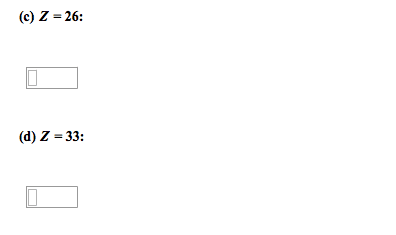CHEM 1A03 Study Guide - Midterm Guide: Alkaline Earth Metal, Alkali Metal, Internal Transcribed Spacer

35
CHEM 1A03 Full Course Notes
Verified Note
35 documents
Document Summary
Metals (left), non-metals (right), metalloids (stairs), noble gases (last column) Exceptions are -o-o- bonds (-1: halogens are typically -1, non metals are typically negatively charged when in a compound with metalloids and metals. Metals are typically positive when with metalloids and non metals which are (-: some elements, like transition metals, have multiple oxidation numbers. Elements with multiple oxidation states are given roman numerals. Molecular: number of molecules must be speci ed (mon, di, tri etc ) Parent molecule (ate), +1 o=per, -1 o= ite, -2 o=hypo-ite, or parent (ic), +1 o=per, -1 o=ous, Multiply by the constant in-front of desired element. Versions of an atom or element which have the same number of protons but a different number of neutrons. When given all isotopes except 1, nd the sum of all fractions and subtract from 1 to nd fraction of a?.